Advertisements
Advertisements
प्रश्न
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
Advertisements
उत्तर
Relation between the change in internal energy (ΔU), work is done (W), and heat (Q):
- When the amount of heat Q is added to the system and the system does not do any work during the process, its internal energy increases by the amount, ΔU = Q.
- When the system does some work to increase its volume, and no heat is added to it while expanding, the system loses energy to its surrounding,s and its internal energy decreases.
∴ ΔU = –W. - As the internal energy can be changed using both ways, we can consider the total change in the internal energy as,
ΔU = Q – W ….(1)
This is the mathematical statement of the first law of thermodynamics.
APPEARS IN
संबंधित प्रश्न
Should the internal energy of a system necessarily increase if its temperature is increased?
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
A force F is applied on a block of mass M. The block is displaced through a distance d in the direction of the force. What is the work done by the force on the block? Does the internal energy change because of this work?
The outer surface of a cylinder containing a gas is rubbed vigorously by a polishing machine. The cylinder and its gas become warm. Is the energy transferred to the gas heat or work?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
The final volume of a system is equal to the initial volume in a certain process. Is the work done by the system necessarily zero? Is it necessarily nonzero?
Consider the following two statements.
(A) If heat is added to a system, its temperature must increase.
(B) If positive work is done by a system in a thermodynamic process, its volume must increase.
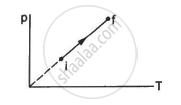
An ideal gas goes from the state i to the state f as shown in figure. The work done by the gas during the process ______________ .
A 100 kg lock is started with a speed of 2.0 m s−1 on a long, rough belt kept fixed in a horizontal position. The coefficient of kinetic friction between the block and the belt is 0.20. (a) Calculate the change in the internal energy of the block-belt system as the block comes to a stop on the belt. (b) Consider the situation from a frame of reference moving at 2.0 m s−1 along the initial velocity of the block. As seen from this frame, the block is gently put on a moving belt and in due time the block starts moving with the belt at 2.0 m s−1. calculate the increase in the kinetic energy of the block as it stops slipping past the belt. (c) Find the work done in this frame by the external force holding the belt.
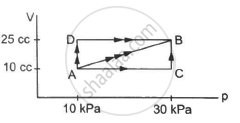
Figure shows three paths through which a gas can be taken from the state A to the state B. Calculate the work done by the gas in each of the three paths.
A substance is taken through the process abc as shown in figure. If the internal energy of the substance increases by 5000 J and a heat of 2625 cal is given to the system, calculate the value of J.

A system releases 130 kJ of heat while 109 kJ of work is done on the system. Calculate the change in internal energy.
What is the energy associated with the random, disordered motion of the molecules of a system called as?
When does a system lose energy to its surroundings and its internal energy decreases?
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
Explain the different ways through which the internal energy of the system can be changed.
The internal energy of a system is ______
An ideal gas is compressed at a constant temperature. Its internal energy will ____________.
When 1 g of water at 0° C and 1 x 105 N/m2 pressure is converted into ice of volume 1.082 cm3, the external work done will be ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
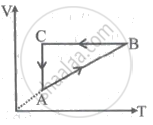
A cyclic process ABCA is shown in the V-T diagram. A process on the P-V diagram is ______.
If a gas is compressed adiabatically:
The internal energy of one mole of argon at 300 K is ______. (R = 8.314 J/mol.K)
A steam engine delivers 4.8 x 108 Jof work per minute and services 1.2 x 109 J of heat per minute from its boiler. What is the percentage efficiency of the engine?
What is heat?
A system releases 125 kJ of heat while 104 kJ work is done on the system. Calculate the change in internal energy.
