Advertisements
Advertisements
प्रश्न
In a process on a system, the initial pressure and volume are equal to the final pressure and volume.
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
(c) The net heat given to the system in the process must be zero.
(d) The net work done by the system in the process must be zero.
Advertisements
उत्तर
(a) The initial temperature must be equal to the final temperature.
(b) The initial internal energy must be equal to the final internal energy.
a) Let initial pressure, volume and temperature be P1, V1 and T1 and final pressure, volume and temperature be P2, V2 and T2. Then,
\[\frac{P_1 V_1}{T_1} = \frac{P_2 V_2}{T_2}\]
\[ \Rightarrow T_1 = T_2...........\left[ P_1 = P_2\text{ and }V_1 = V_2 \right]\]
b) Internal energy is given by ΔU = nCvΔT.
Since ΔT = 0, ΔU = 0.
c) This may not be true because the system may be isothermal and in between, heat may have been given to the system. Also, the system may have done mechanical expansion work and returned back to its original state.
d) Not necessary because an isothermal system may have done work absorbing heat from outside and coming back to the same state losing heat.
APPEARS IN
संबंधित प्रश्न
Explain why Air pressure in a car tyre increases during driving.
Should the internal energy of a system necessarily increase if heat is added to it?
A cylinder containing a gas is lifted from the first floor to the second floor. What is the amount of work done on the gas? What is the amount of work done by the gas? Is the internal energy of the gas increased? Is the temperature of the gas increased?
A force F is applied on a block of mass M. The block is displaced through a distance d in the direction of the force. What is the work done by the force on the block? Does the internal energy change because of this work?
A closed bottle contains some liquid. the bottle is shaken vigorously for 5 minutes. It is found that the temperature of the liquid is increased. Is heat transferred to the liquid? Is work done on the liquid? Neglect expansion on heating.
An ideal gas is pumped into a rigid container having diathermic walls so that the temperature remains constant. In a certain time interval, the pressure in the container is doubled. Is the internal energy of the contents of the container also doubled in the interval ?
When a tyre bursts, the air coming out is cooler than the surrounding air. Explain.
Figure shows two processes A and B on a system. Let ∆Q1 and ∆Q2 be the heat given to the system in processes A and B respectively. Then ____________ .
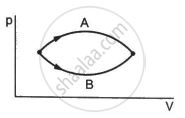
The pressure p and volume V of an ideal gas both increase in a process.
(a) Such a process is not possible.
(b) The work done by the system is positive.
(c) The temperature of the system must increase.
(d) Heat supplied to the gas is equal to the change in internal energy.
Figure shows a cylindrical tube of volume V with adiabatic walls containing an ideal gas. The internal energy of this ideal gas is given by 1.5 nRT. The tube is divided into two equal parts by a fixed diathermic wall. Initially, the pressure and the temperature are p1, T1 on the left and p2, T2 on the right. The system is left for sufficient time so that the temperature becomes equal on the two sides. (a) How much work has been done by the gas on the left part? (b) Find the final pressures on the two sides. (c) Find the final equilibrium temperature. (d) How much heat has flown from the gas on the right to the gas on the left?

What is the energy associated with the random, disordered motion of the molecules of a system called as?
What is the internal energy of the system, when the amount of heat Q is added to the system and the system does not do any work during the process?
A system releases 100 kJ of heat while 80 kJ of work is done on the system. Calculate the change in internal energy.
A cylinder containing one gram molecule of the gas was compressed adiabatically until its temperature rose from 27°C to 97°C. Calculate the work done and heat produced in the gas (𝛾 = 1.5).
derive the relation between the change in internal energy (∆U), work is done (W), and heat (Q).
When 1 g of water at 0° C and 1 x 105 N/m2 pressure is converted into ice of volume 1.082 cm3, the external work done will be ____________.
Two samples A and B, of a gas at the same initial temperature and pressure are compressed from volume V to V/2; A isothermally and B adiabatically. The final pressure of A will be ______.
If a gas is compressed adiabatically:
What is heat?
