Topics
Heredity and Evolution
Life Processes in Living Organisms Part -1
- Life Processes in Living Organisms
- Living Organisms and Energy Production
- Glycolysis
- Tricarboxylic Acid Cycle (Citric Acid Cycle or Kreb’s Cycle)
- Phases of Respiration: Electron Transport Chain (Electron Transfer System)
- Production of ATP
- Cellular Respiration
- Energy From Different Food Components
- Cell Division: an Essential Life Process
- Mitosis > Karyokinesis (Division of Nucleus)
- Mitosis > Cytokinesis (Division of Cytoplasm)
- Significance of Mitosis
- Meiosis: Reduction Division
- Stages of Meiosis: Meiosis I
- Stages of Meiosis: Meiosis II
- Significance of Meiosis
- Mitosis Vs Meiosis
Life Processes in Living Organisms Part - 2
Environmental Management
Towards Green Energy
- Energy and Use of Energy
- Generation of Electrical Energy
- Power Plants Based on Nuclear Energy
- Power Plant Based on Thermal Energy
- Power Plants Based on Natural Gas
- Electric Energy Generation and Environment
- Electricity Generation using Hydroelectric Energy
- Electricity Generation using Wind Energy
- Electricity Generation using Solar Energy
- Solar Photovoltaic Cell
- Solar Thermal Power Plant
Animal Classification
- Classification of Living Organisms
- History of Animal Classification
- Traditional Method of Animal Classification
- Conventional System of Animal Classification
- Criteria for New System of Classification > Grades of Organization
- Criteria for New System of Classification > Body Symmetry
- Criteria for New System of Classification > Germ Layers
- Criteria for New System of Classification > Body cavity (Coelom)
- Criteria for New System of Classification > Body Segmentation
- Phylum: Porifera
- Phylum: Cnidaria/Coelenterata
- Phylum: Platyhelminthes
- Phylum: Aschelminthes
- Phylum: Annelida
- Phylum: Arthropoda
- Phylum: Mollusca
- Phylum: Echinodermata
- Phylum: Hemichordata
- Phylum: Chordata
- Subphylum: Urochordata
- Subphylum: Cephalochordata
- Subphylum: Vertebrata/Craniata
- Subphylum: Vertebrata/Craniata > Class: Cyclostomata
- Subphylum: Vertebrata/Craniata > Class: Pisces
- Subphylum: Vertebrata/Craniata > Class: Amphibia
- Subphylum: Vertebrata/Craniata > Class: Reptilia
- Subphylum: Vertebrata/Craniata > Class: Aves
- Subphylum: Vertebrata/Craniata > Class: Mammalia
Introduction to Microbiology
Cell Biology and Biotechnology
- Cell Biology (Cytology)
- Stem Cells
- Stem Cell Research
- Organ Transplantation
- Organ and Body Donation
- Concept of Biotechnology
- Crop Biotechnology > Hybrid Seeds
- Crop Biotechnology > Genetically Modified Organisms (GMOs)
- Crop Biotechnology > Biofertilizers
- Animal Husbandry (Livestock)
- Human Health
- Vaccination and Immunization
- Edible Vaccines
- Treatment
- Interferons
- Gene Therapy
- Cloning
- Industrial Products/White Biotechnology
- Environment and Biotechnology
- Food Biotechnology
- DNA Fingerprinting
- Green Revolution
- White Revolution
- Blue Revolution
- Fertilizers
- Insecticides
- Types of Farming in India > Organic Farming
- Animal Husbandry (Livestock) > Apiculture (Bee Farming)
- Cultivation of Medicinal Plants
- Processing and Preservation of Fruits
Social Health
Disaster Management
Life's Internal Secrets
- Autotrophic Plants
- Heterotrophic Plants
- Human Digestive System
- Nutrition in Plants
- Transport
The Regulators of Life
The Life Cycle
Mapping Our Genes
Understanding Metals and Non-Metals
Striving for Better Environment 2
- Use of Efficient and Eco-friendly Technology
- Sustainable Use of Resources
- Enforcement of Acts, Laws and Policies
Amazing World of Carbon Compounds
- Definition: Ion
- Definition: Cation
- Definition: Anion
- Definition: Electropositive Element
- Definition: Electronegative Element
- Definition: Electrovalent Bond
- Definition: Electrovalent (or Ionic) Compounds
- Definition: Electrovalency
Introduction of Ionic Bond:
An ionic bond is a type of chemical bond formed through the transfer of electrons from one atom to another. It typically occurs between a metal and a nonmetal.
- In this process, the metal atom loses one or more electrons to become a positively charged ion (cation), while the nonmetal gains these electrons to become a negatively charged ion (anion).
- The electrostatic force of attraction between the oppositely charged ions holds them together in a stable structure, forming an ionic bond.
- Ionic bonds are responsible for the formation of ionic compounds, such as sodium chloride (NaCl).
- These compounds exhibit unique properties, including high melting and boiling points, solubility in water, and the ability to conduct electricity in molten or aqueous states.
- The ionic bond allows atoms to achieve a stable electron configuration, often following the octet rule, which makes them energetically stable.
Formation of Ionic Bonds
(a) Formation of Ionic Bond in NaCl (Sodium Chloride):
- Sodium (Na): Atomic number 11, configuration 2,8,1. It has 1 valence electron in its outermost shell (M shell).
- Chlorine (Cl): Atomic number 17, configuration 2,8,7. It has 7 valence electrons and needs 1 more electron to complete its octet.
- Sodium loses 1 electron, becoming a Na⁺ cation (positively charged ion). New configuration of Na⁺: 2,8 (stable octet).
- Chlorine gains the electron lost by sodium, becoming a Cl⁻ anion (negatively charged ion). New configuration of Cl⁻: 2, 8, 8 (stable octet).
- The oppositely charged ions (Na⁺ and Cl⁻) are held together by the electrostatic force of attraction, forming an ionic bond.
- The compound formed is sodium chloride (NaCl).
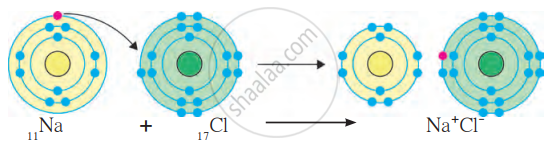
Formation of Ionic bond of NaCl
(b) Formation of Ionic Bond in MgCl₂ (Magnesium Chloride):
- Magnesium (Mg): Atomic number = 12, electronic configuration = 2, 8, 2.
- Magnesium has 2 valence electrons in its outermost shell. It needs to lose 2 electrons to achieve a stable octet configuration.
- Chlorine (Cl): Atomic number = 17, electronic configuration = 2, 8, 7.
- Chlorine has 7 valence electrons in its outermost shell. Each chlorine atom needs 1 electron to complete its octet.
- Magnesium loses 2 electrons, forming a Mg²⁺ cation with a stable configuration of 2, 8.
- Each chlorine atom gains 1 electron from magnesium to form a Cl⁻ anion with a stable configuration of 2, 8, 8.
Mg → Mg²⁺ + 2e⁻
Cl + e⁻ → Cl⁻
Magnesium loses 2 electrons, and these are gained by 2 chlorine atoms.
Mg²⁺ + 2Cl⁻ → MgCl₂
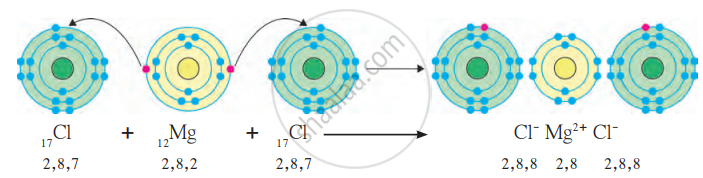
Formation of Ionic bond in MgCl₂ molecule
(c) Formation of Ionic Bond in KF (Potassium Fluoride):
- Potassium (K): Atomic number = 19, electronic configuration = 2, 8, 8, 1.
- Potassium has 1 valence electron in its outermost shell. And it needs to lose 1 electron to achieve a stable octet configuration.
- Fluorine (F): Atomic number = 9, electronic configuration = 2, 7.
- Fluorine has 7 valence electrons in its outermost shell. And it needs to gain 1 electron to complete its octet.
- Potassium loses 1 electron, forming a K⁺ cation with a stable configuration of 2, 8, 8.
- Fluorine gains 1 electron from potassium to form a F⁻ anion with a stable configuration of 2, 8.
K → K⁺ + e⁻
F + e⁻ → F⁻
Potassium loses 1 electron, and fluorine gains 1 electron, ensuring charge balance:
K⁺ + F⁻ → KF

Formation of Ionic bond in KF molecule
(d) Formation of Ionic Bond in CaO (Calcium Oxide):
- Calcium (Ca): Atomic number = 20, electronic configuration = 2, 8, 8, 2.
- Calcium has 2 valence electrons in its outermost shell. And it needs to lose 2 electrons to achieve a stable octet configuration.
- Oxygen (O): Atomic number = 8, electronic configuration = 2, 6.
- Oxygen has 6 valence electrons in its outermost shell. And it needs to gain 2 electrons to complete its octet.
- Calcium loses 2 electrons, forming a Ca²⁺ cation with a stable configuration of 2, 8, 8.
- Oxygen gains 2 electrons from calcium to form an O²⁻ anion with a stable configuration of 2, 8.
Ca → Ca²⁺ + 2e⁻
O + 2e⁻ → O²⁻
Calcium loses 2 electrons, and oxygen gains 2 electrons, ensuring charge balance:

Formation of Ionic bond in CaO molecule
Properties of Ionic Compounds
Melting point and boiling point of some ionic compounds:
| Ionic Compound | Melting Point (K) | Boiling Point (K) |
|---|---|---|
| NaCl | 1074 | 1686 |
| LiCl | 887 | 1600 |
| CaCl₂ | 1045 | 1900 |
| CaO | 2850 | 3120 |
| MgCl₂ | 981 | 1685 |
- Ionic compounds are solid and hard due to the strong electrostatic force between positive and negative ions but are generally brittle, breaking when pressure is applied.
- They have high melting and boiling points because a large amount of energy is needed to overcome the strong ionic bonds.
- Ionic compounds are typically soluble in water, as water's polarity helps separate the ions, but they are insoluble in non-polar solvents like kerosene or petrol.
- In solid form, ionic compounds do not conduct electricity because the ions are fixed in place in the crystal lattice and cannot move.
- In molten form or aqueous solutions, ionic compounds conduct electricity as the ions are free to move and carry electric charge.
- Ionic compounds are characterised by a rigid lattice structure, which provides stability but limits flexibility.
Definition: Ion
An ion is a charged particle which is formed due to the gain or the loss of one or more electrons by an atom.
Definition: Cation
A metallic atom, which loses electron(s), becomes a positively charged ion and is known as a cation.
Definition: Anion
A non-metallic atom, which gains electron(s), becomes a negatively charged ion and is known as an anion.
Definition: Electropositive Element
A metallic element, whose one atom readily loses electron(s) to form a positively charged ion, is an electropositive element.
Definition: Electronegative Element
A non-metallic element, whose atom readily accepts electron(s) to form a negatively charged ion, is an electronegative element.
Definition: Electrovalent Bond
The cation and the anion being oppositely charged attract each other and form a chemical bond. Since this chemical bond formation is due to the electrostatic force of attraction between a cation and an anion, it is called an electrovalent (or an ionic) bond.
Definition: Electrovalent (or Ionic) Compounds
The chemical compounds formed as a result of the transfer of electrons from one atom of an element to one atom of another element are called ionic (or electrovalent) compounds.
Definition: Electrovalency
The number of electrons that an atom of an element loses or gains to form a electrovalent bond is called its electrovalency.
Maharashtra State Board: Class 12
Key Points: Ionic (Electrovalent) Bond
An ionic bond is formed by the complete transfer of one or more electrons from an electropositive atom to an electronegative atom, resulting in oppositely charged ions that attract each other.
Key conditions for ionic bond formation:
-
One atom must have low ionisation enthalpy (easily loses electron) — typically a metal
-
The other must have high electron affinity (easily gains electron) — typically a non-metal
-
Large difference in electronegativity between the two atoms
Example: Na + Cl → Na⁺ + Cl⁻ → NaCl
-
Sodium (2,8,1) loses 1 electron → Na⁺ (2,8)
-
Chlorine (2,8,7) gains 1 electron → Cl⁻ (2,8,8)
Ionic solids are crystalline structures containing cations and anions held together by strong electrostatic ionic bonds.
