Advertisements
Advertisements
Give scientific reasons.
Silver amalgam is used for filling dental cavities.
Concept: Corrosion of Metals
| A process of forming a thick oxide of aluminium when aluminium is exposed to air. This coat makes it resistant to corrosion. Resistance can be improved by making a layer of oxide thinker. In this technique, the aluminium article is the anode, and the electrolyte is sulphuric acid. The anode reaction results in the formation of a black-coloured film of aluminium oxide on the anode. By putting appropriate dyes in the electrolytic solution, both coloured surface with the decorative finish is achieved. Kitchen articles like anodised such as pressure cookers, pans and frames of sliding windows are applications of this technique. |
- Name the anode and electrolyte used in this technique.
- How can we make aluminium articles made resistant to corrosion?
- Name the technique used to coat the aluminium articles.
Concept: Corrosion of Metals
Answer the questions based on the figure below:

- Which reaction is shown in the figure?
- Which gas is evolved in the reaction?
- Give an example of reactants that rapidly show this reaction. Give equations.
- Give an example of reactants that do not react rapidly.
- In what condition will reactants of (c) part react? Give equation.
Concept: Chemical Properties of Metal
In ______ process a layer of molten tin is deposited on metals.
Concept: Corrosion of Metals
Corrosion can be prevented by using ……………. solution.
Concept: Chemical Properties of Carbon Compounds > Oxidation
The chemical formula for rust is.............. .
Concept: Chemical Properties of Carbon Compounds > Oxidation
What is meant by rust?
Concept: Chemical Properties of Carbon Compounds > Oxidation
Write the chemical formula of rust.
Concept: Chemical Properties of Carbon Compounds > Oxidation
Give scientific reason.
It is reccommended to use air tight container for storing oil for long time.
Concept: Chemical Properties of Carbon Compounds > Oxidation
Identify from the reaction the reactants that undergo oxidation and reduction.
Fe + S → FeS
Concept: Chemical Properties of Carbon Compounds > Oxidation
Complete the following flow chart.
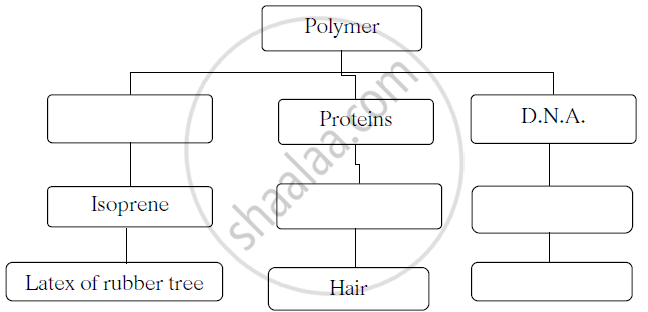
Concept: Macromolecules and Polymers
Concept: Chemical Properties of Carbon Compounds > Oxidation
Complete the correlation:
Alkene : C = C :: Alkyne: _______.
Concept: Homologous Series
Define Hydrocarbons.
Concept: Functional Groups in Carbon Compounds
The unsaturated hydrocarbons containing a carbon-carbon double bond are called _______.
Concept: Homologous Series
The general molecular formula for the homologous series of alkynes is _______.
Concept: Homologous Series
Write the molecular formula of the given compound.
Ethyl ethanoate
Concept: Functional Groups in Carbon Compounds
Cyclohexane is a branched chain type of hydrocarbon.
Concept: Functional Groups in Carbon Compounds
Distinguish between:
Saturated hydrocarbons - Unsaturated hydrocarbons
Concept: Functional Groups in Carbon Compounds
