HSC Science (General)
HSC Science (Electronics)
HSC Science (Computer Science)
Academic Year: 2022-2023
Date & Time: 24th July 2023, 11:00 am
Duration: 3h
Advertisements
General Instructions:
The question paper is divided into four sections:
- Section A: Q. No.: 1 contains Ten multiple choice type of questions carrying One mark each. Q. No. 2 contains Eight very short answer type of questions carrying One mark each.
- Section B: Q. No. 3 to Q. No. 14 contain Twelve short answer type of questions carrying Two marks each. (Attempt any Eight)
- Section C: Q. No. 15 to Q. No. 26 contain Twelve short answer type of questions carrying Three marks each. (Attempt any Eight)
- Section D: Q. No. 27 to Q. No. 31 contain Five long answer type of questions carrying Four marks each. (Attempt any Three)
- Use of the log table is allowed. Use of calculator is not allowed.
- Figures to the right indicate full marks.
- For each multiple choice type of question, it is mandatory to write the correct answer along with its alphabet. e.g. (a) ....../ (b) ....../ (c) ....../ (d) ...... No mark(s) shall be given if ONLY the correct answer or the alphabet of the correct answer is written. Only the first attempt will be considered for evaluation.
- Physical Constants:
Avogadro Number= NA= 6.022 × 1023
Anisole on heating with concentrated HI gives ______.
Iodobenzene
Phenol + Methanol
Phenol + Iodomethane
Iodobenzene + methanol
Chapter: [11] Alcohols, Phenols and Ethers
Which solution shows positive deviation from Raoult’s law?
Phenol and Aniline
Chloroform and Acetone
Ethanol and Acetone
Chloroform and Ethanol
Chapter:
The coordination number of cobalt in [CoCl2(en)2]+ is ______.
6
4
2
0
Chapter:

The name of the above reaction is ______.
Etard reaction
Friedel craft acylation reaction
Stephan reaction
Gatterman-Koch reaction
Chapter:
Which is an example of thermoplastic polymer?
Bakelite
Polystyrene
Nylon 6, 6
Urea formaldehyde resin
Chapter:
Nichrome is an alloy of ______.
Cu, Sn
Cu, Ni
Ni, Cr
Fe, Cr
Chapter:
Identify ‘A’ in the following reaction:
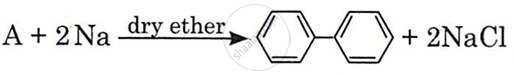
Bromobenzene
1, 4-dichlorobenzene
Naphthalene
Chlorobenzene
Chapter:
Which amine does NOT react with Hinsberg reagent?
Ethanamine
N-ethylethanamine
N, N-diethylethanamine
2-methyl-propan-2-amine
Chapter:
The dissociation constant for NH4OH is 1.8 × 10−5. The degree of dissociation in its 0.01 M solution is ______.
0.04242
0.4242
0.004242
4.242
Chapter:
Half-life of a first order reaction is 30 minutes at 300 K. The value of its rate constant, K is ______.
2.31 min−1
0.0231 min−1
0.231 min−1
2.310 × 10−3 min−1
Chapter:
Write the name of the radioactive element in group 16.
Chapter:
Write the number of particles present in FCC per unit cell.
Chapter:
Name the γ-isomer of BHC.
Chapter: [16] Green Chemistry and Nanochemistry
Which alloy is used in the Fischer-Tropsch process in the synthesis of gasoline?
Chapter: [8] Transition and Inner Transition Elements
Three moles of an ideal gas are expanded isothermally from 15 dm3 to 20 dm3 at a constant external pressure of 1.2 bar. Calculate the amount of work in Joules.
Chapter: [4] Chemical Thermodynamics
Write postulates of Werner’s theory of co-ordination complexes.
Chapter: [9] Coordination Compounds
Why does fluorine show anomalous behaviour in ‘17 group’ elements?
Chapter:
What is the mass of Cu metal produced at the cathode during the passage of 5 ampere current through CuSO4 solution for 6000 seconds? Molar mass of Cu is 63.5 g mol−1.
Chapter:
How is glucose prepared from cane sugar (sucrose)?
Chapter: [14.01] Carbohydrates
Derive integrated rate law for zero order reaction.
Chapter: [6] Chemical Kinetics
The normal boiling point of ethyl acetate is 77.06°C. A solution of 50 g of a non-volatile solute in 150 g of ethyl acetate boils at 84.27°C. Evaluate the molar mass of solute if Kb for ethyl acetate is 2.77°C kg mo1−1.
Chapter:
How is phenol prepared from cumene?
Chapter: [11.02] Phenols
Why do the d-block elements form coloured compounds?
Chapter: [8.01] D-block Elements
Advertisements
Write a note on the Wolf-Kishner reduction reaction.
Chapter:
Write the preparation of nylon 6, 6.
Chapter: [15] Introduction to Polymer Chemistry
Calculate the standard enthalpy of the reaction, \[\ce{SiO2_{(s)} + 3C_{(graphite)} -> SiC_{(s)} + 2CO_{(g)}}\] from the following reactions:
- \[\ce{Si_{(s)} + O2_{(g)} -> SiO2_{(s)}}\], ΔrH0 = −911 kJ
- \[\ce{2C_{(graphite)} + O2_{(g)} -> 2CO_{(g)}}\], ΔrH0 = −221 kJ
- \[\ce{Si_{(s)} + C_{(graphite)} -> SiC_{(s)}}\], ΔrH0 = −65.3 kJ
Chapter: [4] Chemical Thermodynamics
Write a short note on the following:
Hoffmann’s bromamide reaction
Chapter: [13] Amines [13.01] Amines
Write any three advantages of nanoparticles and nanotechnology.
Chapter: [16] Green Chemistry and Nanochemistry
State disadvantages of nanoparticles and nanotechnology.
Chapter: [16] Green Chemistry and Nanochemistry
Write the molecular formula and structure of sulphuric acid.
Chapter:
Write the molecular formula and structures of the peroxy monosulphuric acid.
Chapter: [7.02] Group 16 Elements
Write the molecular formula and structure of thiosulphuric acid.
Chapter:
Explain optical activity of 2-chlorobutane.
Chapter: [10] Halogen Derivatives
Write the different oxidation states of manganese.
Chapter: [8.01] D-block Elements
Why +2 oxidation state of manganese is more stable?
Chapter: [8.01] D-block Elements
Write the preparation of ethanol from methyl magnesium iodide.
Chapter: [11] Alcohols, Phenols and Ethers
Prepare the following by using methyl magnesium iodide:
Propan-2-ol
Chapter:
Prepare the following by using methyl magnesium iodide:
2-methylpropan-2-ol
Chapter:
Derive the relation between freezing point depression and molar mass of solute.
Chapter: [2] Solutions
Advertisements
Write any four applications of buffer solution.
Chapter: [3] Ionic Equilibria
An element with molar mass 27 g/mol forms a cubic unit cell with edge length of 405 p.m. If the density of the element is 2.7 g/cm3, what is the nature of the cubic unit cell?
Chapter: [1] Solid State
On the basis of valence bond theory, explain the nature of bonding in the [Ni(Cl)4]2− complex ion.
Chapter:
How will you convert sodium acetate to methane?
Chapter: [12.01] Aldehydes and Ketones
Write Arrhenius equation and explains the terms involved.
Chapter:
How will you determine activation energy from rate constants at two different temperatures?
Chapter: [6] Chemical Kinetics
What are interhalogen compounds?
Chapter: [7] Elements of Groups 16, 17 and 18
Write any two characteristics of interhalogen compounds.
Chapter: [7] Elements of Groups 16, 17 and 18
Draw the Fischer projection formula for α-D-(t) glucose.
Chapter:
Write the reactions involved in the preparation of Teflon.
Chapter: [15] Introduction to Polymer Chemistry [15] Polymers
Describe the construction and working of Standard Hydrogen Electrode.
Chapter:
Write any two difficulties of setting SHE (Standard Hydrogen Electrode).
Chapter: [5] Electrochemistry
Write any two statements of the first law of thermodynamics.
Chapter:
For a certain reaction ΔH0 is −224 kJ and ΔS0 is −153 J K−1. At what temperature the change over from spontaneous to non-spontaneous will occur?
Chapter: [4] Chemical Thermodynamics
Write the environmental effect of methylene chloride.
Chapter:
Other Solutions
Submit Question Paper
Help us maintain new question papers on Shaalaa.com, so we can continue to help studentsonly jpg, png and pdf files
Maharashtra State Board previous year question papers 12th Standard Board Exam Chemistry with solutions 2022 - 2023
Previous year Question paper for Maharashtra State Board 12th Standard Board Exam Chemistry-2023 is solved by experts. Solved question papers gives you the chance to check yourself after your mock test.
By referring the question paper Solutions for Chemistry, you can scale your preparation level and work on your weak areas. It will also help the candidates in developing the time-management skills. Practice makes perfect, and there is no better way to practice than to attempt previous year question paper solutions of Maharashtra State Board 12th Standard Board Exam.
How Maharashtra State Board 12th Standard Board Exam Question Paper solutions Help Students ?
• Question paper solutions for Chemistry will helps students to prepare for exam.
• Question paper with answer will boost students confidence in exam time and also give you an idea About the important questions and topics to be prepared for the board exam.
• For finding solution of question papers no need to refer so multiple sources like textbook or guides.
