Advertisements
Advertisements
प्रश्न
Write two uses and two properties of polythene.
Advertisements
उत्तर
Uses of high-density polyethylene:
- HDP is used in the manufacture of toys and other household articles like buckets, dustbins, bottles, pipes, etc.
- It is used to prepare laboratory wares and other objects where high tensile strength and stiffness is required.
Properties of high-density polyethylene:
- HDP is crystalline, melting point in the range of 144 - 150 °C.
- It is much stiffer than LDP and has high tensile strength and hardness.
- It is more resistant to chemicals than LDP.
APPEARS IN
संबंधित प्रश्न
Write the reactions involved in the preparation of PVC
Write the chemical reaction to prepare novolac polymer.
Explain the following term: Homopolymers
Based on molecular forces, what type of polymer is neoprene?
Write the formulae of the raw materials used for preparation of Dextran.
Draw the structures of veronal and thymine.
Answer the following in one sentence.
Identify 'A' in the following reaction:
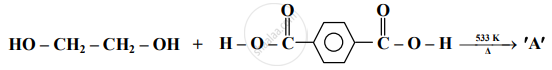
Answer the following in one sentence.
Identify 'B' in the following reaction:
\[\ce{H2N -(CH2)6 - NH2 + HOOC - (CH2)4 - COOH ->[N2][533 K]}\]'B'
Answer the following in one sentence.
Identify thermoplastic and thermosetting plastic from the following:
- PET
- Urea formaldehyde resin
- Polythene
- Phenol formaldehyde resin
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Identify condensation polymers and addition polymers from the following.
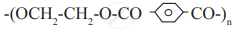
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Write preparation, properties, and uses of Teflon.
Answer the following.
Is synthetic rubber better than natural rubber? If so, in what respect?
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Monomer used for preparation of polyacrylonitrile is _____________
Write chemical reaction for preparation of the following.
Buna-S
Write the name and formulae of the monomers used for the preparation of dacron.
Explain vulcanization of rubber.
Which of the following is used as a substitute for wool?
Which of the following polymer is used in paints?
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
How many isoprene units are present in abscisic acid?
Which of the following is the monomer of neoprene?
Select the CORRECT match for both the polymers.
\[\ce{{n} CH2 = CH2 ->[333 K - 343 K][6 - 7 atm, catalyst] X}\]
Which of the following is CORRECT about polymer 'X'?
Novolac is obtained from ____________.
Which of the following compounds is used to prepare orlon?
Which of the following polymers is prepared by using phenol?
Which of the following pair of compounds is used as monomers for bakelite?
Which of the following polymers is used as insulation for cables?
Which among the following is an example of addition polymer?
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Name the polymers used in laminated sheets and give the name of monomeric units involved in its formation.
Match the polymers given in Column I with the type of linkage present in them given in Column II.
| Column I | Column II |
| (i) Terylene | (a) Glycosidic linkage |
| (ii) Nylon | (b) Ester linkage |
| (iii) Cellulose | (c) Phosphodiester linkage |
| (iv) Protein | (d) Amide linkage |
| v) RNA |
Which one of the following polymers are prepared by addition polymerization?
Nylon threads are made of ______.
Which of the following polymers do not involve cross linkages?
Which of the following is a polymer of enzyme?
Answer the following.
Write the structure of isoprene and the polymer obtained from it.
Write the preparation of viscose rayon.
Write the structure of isoprene and the polymer obtained from it.
Write the name of the monomer of natural rubber.
Another name of terylene is ______.
Write the structure and name of monomer of Natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
