Advertisements
Advertisements
प्रश्न
Write the reaction involved in the formation of bakelite.
How are the following prepared?
Bakelite
How is bakelite made?
How is bakelite synthesised?
Advertisements
उत्तर १
Step 1:

Step 2:

Step 3:

उत्तर २
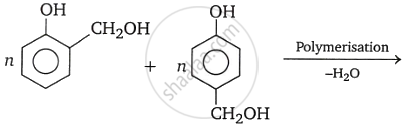
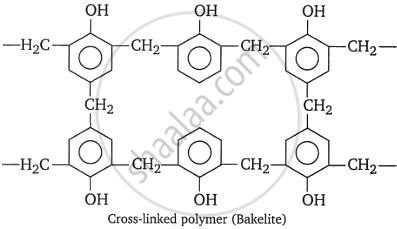
When formaldehyde and phenol react with a basic catalyst present, phenol-formaldehyde resins are created. Methylene bridges can develop at ortho, para, or both ortho and para locations during the process. This allows for the preparation of phenol-formaldehyde resin, often known as bakelite, a linear or cross-linked polymer.
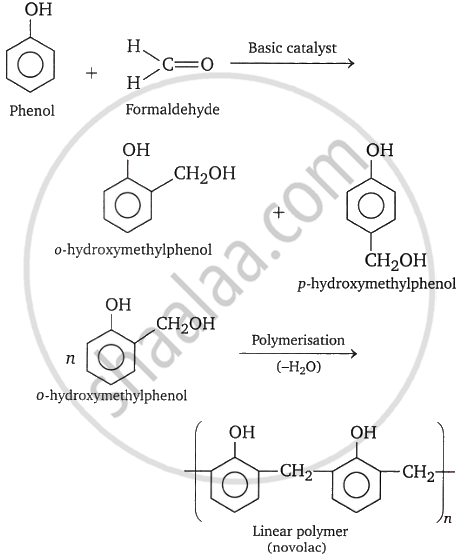
Novolac is a linear polymer used in paints. Novolac undergoes cross-linking when heated with formaldehyde, forming an infusible solid mass known as Bakelite.
Notes
Students should refer to the answer according to their questions.
APPEARS IN
संबंधित प्रश्न
Write any ‘two’ uses of terylene.
Write the chemical reaction to prepare novolac polymer.
Based on molecular forces, what type of polymer is neoprene?
Write the monomers of the following polymer :

Answer the following in one sentence.
Identify 'B' in the following reaction:
\[\ce{H2N -(CH2)6 - NH2 + HOOC - (CH2)4 - COOH ->[N2][533 K]}\]'B'
Answer the following in one sentence.
What type of intermolecular force leads to high-density polymer?
Answer the following.
Write the reaction of the formation of Nylon 6.
Answer the following.
Write the reaction of the formation of Terylene.
Draw the structures of polymers formed from the following monomers
\[\ce{n HOOC–R–COOH + n HO–R'–OH}\]
Identify condensation polymers and addition polymers from the following.
-(CH2 - CH = CH - CH2 -)n
Attempt the following:
Write the chemical reactions involved in the manufacture of Nylon 6,6.
Attempt the following:
What is meant by LDP and HDP? Mention the basic difference between the same with suitable examples.
Answer the following.
Write main specialities of Buna-S, Neoprene rubber?
Monomers ethylene glycol and terephthalic acid undergo condensation polymerization to give polymer called ___________
Monomer of natural rubber is __________
Write a chemical reaction for the preparation of the following polymer.
polyacrylonitrile
Mention two uses of LDP.
Write two uses and two properties of polythene.
Explain vulcanization of rubber.
Which of the following is used as a substitute for wool?
The following structure represents the polymer:
\[\begin{array}{cc}
\ce{[-C-CH2-NH-C-(-CH2)5 NH -]_{{n}}}\\
\phantom{}||\phantom{.............}||\phantom{................}\\
\phantom{}\ce{O}\phantom{.............}\ce{O}\phantom{................}
\end{array}\]
Identify additional polymers from the following.
I. \[\begin{array}{cc}
\ce{-(CH2 - CH -)_{{n}}}\\
\phantom{....}|\\
\phantom{.......}\ce{C6H5}
\end{array}\]
II. \[\ce{-(CH2 - CH = CH - CH2 -)_{{n}}}\]
III. \[\ce{-(CO(CH2)4 - CONH(CH2)6NH -)_{{n}}}\]
IV.
![]()
Which of the following polymers is obtained from chloroprene?
Which of the following compounds is used to prepare orlon?
Which among the following polymers is obtained from CH2 = CH – CN by polymerisation?
Which among the following polymers is an example of addition polymer?
Identify addition polymer from the following.
Which among the following is an example of addition polymer?
Which of the following is not a semisynthetic polymer?
Which of the following polymers, need atleast one diene monomer for their preparation?
(i) Dacron
(ii) Buna-S
(iii) Neoprene
(iv) Novolac
Match the polymers given in Column I with the type of linkage present in them given in Column II.
| Column I | Column II |
| (i) Terylene | (a) Glycosidic linkage |
| (ii) Nylon | (b) Ester linkage |
| (iii) Cellulose | (c) Phosphodiester linkage |
| (iv) Protein | (d) Amide linkage |
| v) RNA |
Which of the following polymer has ester linkage?
Which of the following polymers is synthesized using a free radical polymerisation technique?
Nylon threads are made of ______.
Which of the following polymers do not involve cross linkages?
The catalyst used for the polymerisation of olefins is ______.
Which among the following polymers has high tensile strength and is used to obtain tyre cords?
Which of the foolowing polymer is used in the manufacture of insulators.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Answer the following.
Name and draw structure of the repeating unit in natural rubber.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the name of the monomer of natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Name and draw the structure of the repeating unit in natural rubber.
Name and draw structure of the repeating unit in natural rubber.
Write the structure of isoprene and the polymer obtained from it.
Write the structure of isoprene and the polymer obtained from it.
Which from following statements is NOT true regarding neoprene?
Which among the following is a monomer of natural rubber?
