Advertisements
Advertisements
प्रश्न
Two photons of
विकल्प
equal wavelength have equal linear momenta
equal energies have equal linear momenta
equal frequencies have equal linear momenta
equal linear momenta have equal wavelengths
Advertisements
उत्तर
equal linear momenta have equal wavelengths
Two photons having equal linear momenta have equal wavelengths is correct. As in the rest of the options magnitude of momentum or energy can be same because energy and momentum are inversely proportional to wavelength. But the direction of propagation of the photons can be different.
Hence the correct option is D.
APPEARS IN
संबंधित प्रश्न
The photoelectric cut-off voltage in a certain experiment is 1.5 V. What is the maximum kinetic energy of photoelectrons emitted?
(a) Estimate the speed with which electrons emitted from a heated emitter of an evacuated tube impinge on the collector maintained at a potential difference of 500 V with respect to the emitter. Ignore the small initial speeds of the electrons. The specific charge of the electron, i.e., its e/m is given to be 1.76 × 1011 C kg−1.
(b) Use the same formula you employ in (a) to obtain electron speed for an collector potential of 10 MV. Do you see what is wrong? In what way is the formula to be modified?
Light of intensity 10−5 W m−2 falls on a sodium photo-cell of surface area 2 cm2. Assuming that the top 5 layers of sodium absorb the incident energy, estimate time required for photoelectric emission in the wave-picture of radiation. The work function for the metal is given to be about 2 eV. What is the implication of your answer?
Draw graphs showing variation of photoelectric current with applied voltage for two incident radiations of equal frequency and different intensities. Mark the graph for the radiation of higher intensity.
What is the speed of a photon with respect to another photon if (a) the two photons are going in the same direction and (b) they are going in opposite directions?
Planck's constant has the same dimensions as
Light of wavelength λ falls on a metal with work-function hc/λ0. Photoelectric effect will take place only if
A point source of light is used in a photoelectric effect. If the source is removed farther from the emitting metal, the stopping potential
In an experiment on photoelectric effect, the stopping potential is measured for monochromatic light beams corresponding to different wavelengths. The data collected are as follows:-
Wavelength (nm): 350 400 450 500 550
Stopping potential (V): 1.45 1.00 0.66 0.38 0.16
Plot the stopping potential against inverse of wavelength (1/λ) on a graph paper and find (a) Planck's constant (b) the work function of the emitter and (c) the threshold wavelength.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
The electric field associated with a light wave is given by `E = E_0 sin [(1.57 xx 10^7 "m"^-1)(x - ct)]`. Find the stopping potential when this light is used in an experiment on photoelectric effect with the emitter having work function 1.9 eV.
(Use h = 6.63 × 10-34J-s = 4.14 × 10-15 eV-s, c = 3 × 108 m/s and me = 9.1 × 10-31kg)
Define the term: stopping potential in the photoelectric effect.
Explain how does (i) photoelectric current and (ii) kinetic energy of the photoelectrons emitted in a photocell vary if the frequency of incident radiation is doubled, but keeping the intensity same?
Show the graphical variation in the above two cases.
In photoelectric effect, the photoelectric current started to flow. This means that the frequency of incident radiations is ______.
In photoelectric effect the photo current ______.
Do all the electrons that absorb a photon come out as photoelectrons?
The work function for a metal surface is 4.14 eV. The threshold wavelength for this metal surface is ______.
The graph shows the variation of photocurrent for a photosensitive metal
- What does X and A on the horizontal axis represent?
- Draw this graph for three different values of frequencies of incident radiation ʋ1, ʋ2 and ʋ3 (ʋ3 > ʋ2 > ʋ1) for the same intensity.
- Draw this graph for three different values of intensities of incident radiation I1, I2 and I3 (I3 > I2 > I1) having the same frequency.
- Assertion (A): For the radiation of a frequency greater than the threshold frequency, the photoelectric current is proportional to the intensity of the radiation.
- Reason (R): Greater the number of energy quanta available, the greater the number of electrons absorbing the energy quanta and the greater the number of electrons coming out of the metal.
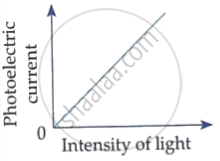
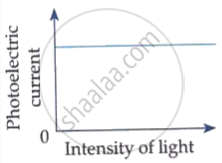
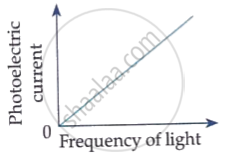
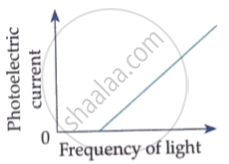
Which of the following options represents the variation of photoelectric current with property of light shown on the x-axis?
The difference between threshold wavelengths for two metal surfaces A and B having work function ΦA = 9 eV and ΦB = 4.5 eV in nm is ______.
(Given, hc = 1242 eV nm)