Advertisements
Advertisements
प्रश्न
Draw the structure of the pyran.
Advertisements
उत्तर

APPEARS IN
संबंधित प्रश्न
Give two evidences for presence of formyl group in glucose.
Draw a neat diagram for the following:
Haworth formula of glucopyranose.
Draw the structure of α-D glucopyranose.
Define carbohydrates.
Write a chemical reaction to convert glucose into glucose cyanohydrin.
By which of the following process formation of glycosidic bond occurs?
Identify the bond that links the carbon of two adjacent monosaccharides.
Which of the following carbohydrate is sweet to taste?
Formation of lactose by conden ation results in the release of how many molecules of water?
From the following identify the group that is exclusively consists of polysaccharides.
From the following which is a heteropolysaccharide?
Identify a non-reducing carbohydrate from the following.
From the following identify the two types of glucose polymers present in starch.
Match the columns and select the correct option.
| Column I | Column II | ||
| i. | Starch | a. | Animal storage molecule |
| ii. | Cellulose | b. | Plant storage molecule |
| iii. | Glycogen | c. | Heparin |
| iv. | Heteropolysaccharide | d. | Plant cell wall component |
4-O-(α-D-Glucopyranosyl)-D-glucopyranose is ____________.
Which one of the following is a carbohydrate but does NOT follow the general formula of carbohydrate?
Which one of the following is NOT soluble in water?
Which following reagent is used to detect presence of five hydroxyl groups in a glucose molecule?
Which among the following statements is true for amylose?
What is the number of hydroxyl groups present in lactic acid?
Identify the number of secondary carbon atoms in glucose.
Which among the following reagents is used to confirm the presence of carbonyl group in glucose?
Identify the number of oxygen atoms present in saccharic acid?
What is the molecular formula of glyceraldehyde?
Which among the following reagents is used to obtain gluconic acid from glucose?
How many hydroxyl groups are present in Erythrulose?
Which among the following observations suggests that glucose also exists in cyclic form?
Which one of the following rotates the plane polarized light towards left?
The correct corresponding order of names of four aldoses with configuration given below Respectively is:
The number of sp2 and sp3 hybridised carbon in fructose are respectively ____________.
Why carbohydrates are generally optically active?
Classify the following into monosaccharides, oligosaccharides and polysaccharides.
Starch
Write the structure of α-D (+) glucopyranose.
Is the following sugar, D-sugar or L-sugar?

A molecule of stachyose contains how many carbon atoms?
Match the Column I with Column II and choose the correct answer from options below:
| Column I | Column II |
| A. Purine | 1. Glycogen |
| B. Pyrimidine | 2. Cellulose |
| C. Structural polysaccharide | 3. Glucagon |
| D. Storage polysaccharide | 4. Adenine |
| 5. Cytosine |
Identify the monosaccharide containing only one asymmetric carbon atom in its molecule.
Which one is a non-reducing commercial sugar?
\[\ce{CH2OH-CO-(CHOH)4-CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Assign D/L configuration to the following monosaccharides:
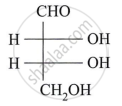
Describe the action of the following reagent on glucose:
Bromine water
Carbohydrates that do not undergo hydrolysis further are called ______.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active?
\[\ce{CH2OH - CO - (CHOH)4 - CH2OH}\] is an example of ______.
Why carbohydrates are generally optically active.
Why carbohydrates are generally optically active.
Two monosaccharides are held together by ______ bond.
Which of the following is used to separate glucose and fructose from hydrolysate of sucrose?
