Advertisements
Advertisements
प्रश्न
Balance the following ionic equations.
\[\ce{Cr2O^{2-}7 + H^{+} + I- -> Cr^{3+} + I2 + H2O}\]
Advertisements
उत्तर
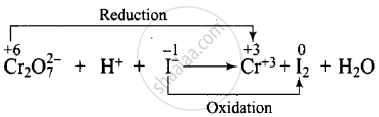
Dividing the equation into two half-reactions:
Oxidation half-reaction: \[\ce{I- -> I2}\]
Reduction half-reaction: \[\ce{CrO7^{2-} -> Cr^{3+}}\]
Balancing oxidation and reduction half-reactions separately as:
Oxidation half-reaction:
\[\ce{I- -> I2}\]
\[\ce{2I- -> I2}\]
\[\ce{2I- -> I2 + 2e-}\] ......(i)
Reduction half-reaction:
\[\ce{CrO^{2-}7 -> Cr^{3+}}\]
\[\ce{Cr2O^{2-}7 -> 2Cr^{3+}}\]
\[\ce{Cr2O^{2-}7 + 6e- -> 2Cr^{3+}}\]
\[\ce{Cr2O^{2-}7 + 14H^{+} + 6e- -> \underset{(acidic medium)}{2Cr^{3+} + 7H2O}}\] .....(ii)
To balance the electrons, multiply equation (i) by 3 and add to equation (ii)
\[\ce{Cr2O^{2-}7 + 14H^{+} + 6I- -> 2Cr^{3+} + 3I2 + 7H2O}\]
APPEARS IN
संबंधित प्रश्न
Calculate the oxidation number of sulphur, chromium and nitrogen in H2SO5, `"Cr"_2"O"_7^(2-)` and `"NO"_3^-`. Suggest structure of these compounds. Count for the fallacy.
The compound AgF2 is an unstable compound. However, if formed, the compound acts as a very strong oxidizing agent. Why?
How do you count for the following observations?
Though alkaline potassium permanganate and acidic potassium permanganate both are used as oxidants, yet in the manufacture of benzoic acid from toluene we use alcoholic potassium permanganate as an oxidant. Why? Write a balanced redox equation for the reaction.
Balance the following equation in basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{P4(s) + OH–(aq) —> PH3(g) + HPO^–_2(aq)}\]
Balance the following equation in basic medium by ion-electron method and oxidation number methods and identify the oxidising agent and the reducing agent.
\[\ce{Cl_2O_{7(g)} + H_2O_{2(aq)} -> ClO-_{2(aq)} + O_{2(g)} + H+_{(aq)}}\]
Balance the following reaction by oxidation number method.
\[\ce{Cr2O^2-_{7(aq)} + SO^2-_{3(aq)}->Cr^3+_{ (aq)} + SO^2-_{4(aq)}(acidic)}\]
Balance the following reaction by oxidation number method.
\[\ce{MnO^-_{4(aq)} + Br^-_{ (aq)}->MnO2_{ (s)} + BrO^-_{3(aq)}(basic)}\]
Identify coefficients 'x' and 'y' for the following reaction.
\[\ce{{x}H2O2_{(aq)} + ClO^-_{4(aq)} -> 2O2_{(g)} + ClO^-_{2(aq)} + {y}H2O_{(l)}}\]
Which of the following is a redox reaction?
Consider the reaction:
\[\ce{6 CO2(g) + 6H2O(l) → C6 H12O6(aq) + 6O2(g)}\]
Why it is more appropriate to write these reaction as:
\[\ce{6CO2(g) + 12H2O(l) → C6 H12O6(aq) + 6H2O(l) + 6O2(g)}\]
Also, suggest a technique to investigate the path of the redox reactions.
Write balanced chemical equation for the following reactions:
Permanganate ion \[\ce{(MnO^{-}4)}\] reacts with sulphur dioxide gas in acidic medium to produce \[\ce{Mn^{2+}}\] and hydrogen sulphate ion.
Balance the following equations by the oxidation number method.
\[\ce{Fe^{2+} + H^{+} + Cr2O^{2-}7 -> Cr^{3+} + Fe^{3+} + H2O}\]
Balance the following equations by the oxidation number method.
\[\ce{I2 + NO^{-}3 -> NO2 + IO^{-}3}\]
Balance the following equations by the oxidation number method.
\[\ce{I2 + S2O^{2-}3 -> I- + S4O^{2-}6}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{HgCl2 (aq) + 2KI (aq) -> HgI2 (s) + 2KCl (aq)}\]
Identify the redox reactions out of the following reactions and identify the oxidising and reducing agents in them.
\[\ce{Fe2O3 (s) + 3CO (g) ->[Δ] 2Fe (s) + 3CO2 (g)}\]
Balance the following ionic equations.
\[\ce{MnO^{-}4 + H^{+} + Br^{-} -> Mn^{2+} + Br2 + H2O}\]
In acidic medium, reaction, \[\ce{MNO^-_4 → Mn^2+}\] an example of ____________.
In the reaction of oxalate with permanganate in an acidic medium, the number of electrons involved in producing one molecule of CO2 is ______.
\[\ce{H2O2 -> 2H^+ + O2 + 2e^-}\]; E0 = −0.68 V.
This equation represents which of the following behaviour of H2O2?
