Advertisements
Advertisements
प्रश्न
At constant temperature, the effect of change of pressure on the volume of a gas was as given below:
|
Pressure in atmosphere |
Volume in liters |
|
0.20 |
112 |
|
0.25 |
89.2 |
|
0.40 |
56.25 |
|
0.60 |
37.40 |
|
0.80 |
28.10 |
|
1.00 |
22.4 |
(a) Plot the following graphs
- P vs V
- P vs 1/V
- PV vs P
Interpret each graph in terms of the law.
(b) Assuming that the pressure values given above are correct, find the correct measurement of the volume.
Advertisements
उत्तर
|
P/atm |
V/dm3 |
1/V |
PV |
|
0.2 |
112 |
0.009 |
22.4 |
|
0.25 |
89.2 |
0.011 |
22.4 |
|
0.4 |
56.25 |
0.018 |
22.4 |
|
0.6 |
37.4 |
0.027 |
22.4 |
|
0.8 |
28.1 |
0.036 |
22.4 |
|
1 |
22.4 |
0.045 |
22.4 |
(a) Plot the following graphs:
1. P vs. V:
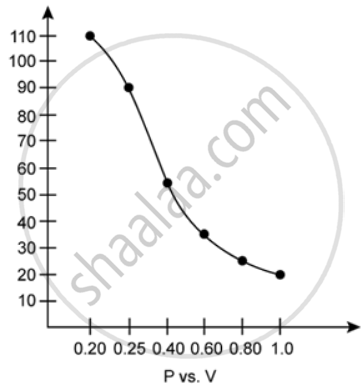
At constant temperature, P is inversely proportional to V. Thus, the plot of V versus P will be a rectangular hyperbola.
2. P vs. 1/V:
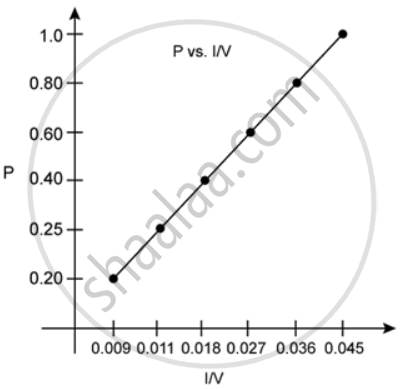
According to Boyle's law, at a constant temperature, the pressure of a fixed amount of gas varies inversely to its volume. The graph of pressure versus 1/V shows a positive slope.
3. PV vs. P:

According to Boyle's law, the product of pressure and volume is constant at a constant temperature. The graph of PV versus P is constant which indicates that the given gas obeys Boyle's law.
(b) The correct measurements of the volume are given below:
|
P/atm |
V/dm3 |
|
0.2 |
112 |
|
0.25 |
89.6 |
|
0.4 |
56 |
|
0.6 |
37.33 |
|
0.8 |
28 |
|
1 |
22.4 |
APPEARS IN
संबंधित प्रश्न
Explain Boyle's Law on the basis of the kinetic theory of matter.
A certain amount of a gas occupies a volume of 0.4 litre at 17°C. To what temperature should it be heated so that its volume gets (a) doubled, (b) reduced to half, pressure remaining constant?
A certain mass of a gas occupies 2 litres at 27°C and 100 Pa. Find the temperature when volume and pressure become half of their initial values.
Calculate the volume occupied by 2 g of hydrogen at 27°C and 4-atmosphere pressure if at STP it occupies 22.4 litres.
Calculate the volume of dry air at STP that occupies 28 cm3 at 14°C and 750 mmHg pressure when saturated with water vapour. The vapour pressure of water at 14°C is 12 mmHg.
Calculate the following:
A gas ‘X’ is collected over water at 17°C and 750 mm. pressure. If the volume of the gas collected is 50 cc., calculate the volume of the dry gas at s.t.p. [at 17°C the vapour pressure is 14 mm.]
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1800 ml. originally occupying 300 ml. at 6 atms. pressure.
Assuming temperature remaining constant calculate the pressure of the gas in the following:
The pressure of a gas having volume 1500 cm3 originally occupying 750 cc. at 5 ats. pressure.
Calculate the coefficient of cubical expansion of a zinc bar. Whose volume is increased 0.25 m3 from 0.3 m3 due to the change in its temperature of 50K?
The following question refers to one mole of chlorine gas.
What will happen to volume of gas, if pressure is doubled?
