Advertisements
Advertisements
Write a short note on the following:
Coupling reaction
Concept: undefined >> undefined
CH3CO2H or CH2FCO2H
Concept: undefined >> undefined
Advertisements
Which acid of the pair shown here would you expect to be stronger?
CH2FCO2H or CH2ClCO2H
Concept: undefined >> undefined
Which acid of the pair shown here would you expect to be stronger?
CH2FCH2CH2CO2H or CH3CHFCH2CO2H
Concept: undefined >> undefined
Which acid of the pair shown here would you expect to be stronger?
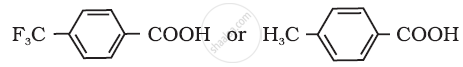
Concept: undefined >> undefined
Arrange the following compounds in increasing order of their property as indicated:
CH3CH2CH(Br)COOH, CH3CH(Br)CH2COOH, (CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)
Concept: undefined >> undefined
Arrange the following compounds in increasing order of their property as indicated:
Benzoic acid, 4-Nitrobenzoic acid, 3, 4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)
Concept: undefined >> undefined
Mention three areas in which H2SO4 plays an important role.
Concept: undefined >> undefined
Why is `K_(a_2) "<<" K_(a_1)` for `H_2SO_4` in water?
Concept: undefined >> undefined
Describe the manufacture of H2SO4 by contact process?
Concept: undefined >> undefined
Write balanced equations for NaCl is heated with sulphuric acid in the presence of MnO2
Concept: undefined >> undefined
Iron (z=26) is highly ferromagnetic. Explain.
Concept: undefined >> undefined
What type of magnetism is shown in the following alignment of magnetic moments?

Concept: undefined >> undefined
Account for the following: Fluorine does not exhibit positive oxidation state.
Concept: undefined >> undefined
Write the major products(s) in the following:

Concept: undefined >> undefined
Name the reagents used in the following reactions:

Concept: undefined >> undefined
Predict the products of the following reactions:

Concept: undefined >> undefined
Fluorine is a stronger oxidising agent than chlorine. Why?
Concept: undefined >> undefined
What type of magnetism is shown by a substance if magnetic moments of domains are arranged in same direction?
Concept: undefined >> undefined
