Advertisements
Advertisements
Question
Describe the manufacture of H2SO4 by contact process?
Advertisements
Solution 1
Sulphuric acid is manufactured by the contact process. It involves the following steps:
Step (i):
Sulphur or sulphide ores are burnt in air to form SO2.
Step (ii):
By a reaction with oxygen, SO2 is converted into SO3 in the presence of V2O5 as a catalyst.
2SO2(g) + O2(g) 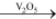 2SO3(g)
2SO3(g)
Step (iii):
SO3 produced is absorbed on H2SO4 to give H2S2O7 (oleum).
`SO_3+H_2SO_4 -> H_2S_2O_7`
This oleum is then diluted to obtain H2SO4 of the desired concentration.
In practice, the plant is operated at 2 bar (pressure) and 720 K (temperature). The sulphuric acid thus obtained is 96-98% pure
Solution 2
Preparation of sulphuric acid:By Contact Process: Burning of sulphur or sulphide ores in presence of oxygen to produce SO2. Catalytic oxidation of SO2 with O2to give SO3 in the presence of V2O5.
2SO2(g) + O2(g) 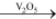 2SO3(g)
2SO3(g)
Then SO3 made to react with sulphuric acid of suitable normality to obtain a thick oily liquid called oleum.
`SO_3(g) + H_2SO_4(l) -> H_2S_2O_7(l)`
Then oleum is diluted to obtain sulphuric acid of desired concentration
`H_2S_2O_7(l) + H_2O(l) -> 2H_2SO_4(l)`
The sulphuric acid obtained by contact process is 96-98% pure
RELATED QUESTIONS
What is molecular formula of oleum?
(a) H2SO3
(b) H2SO4
(c) H2S2O7
(d) H2S2O8
Write the conditions to maximize the yield of H2SO4 by contact process.
Complete the following equations: CaF2+H2SO4 →
Complete the following equations: C + conc. H2SO4 →
Draw the structure of the following:
H2SO4
Mention three areas in which H2SO4 plays an important role.
Write chemical reactions for different steps in the manufacture of sulphuric acid by lead chamber process. Draw the structure of phosphorous pentachloride
Complete and balance the following equations:
S+H2SO4(conc.) →
Write a balanced chemical equation for the following reaction:
Sulphuric acid is treated with phosphorous.
Which of the following reaction is NOT involved in contact process used for manufacturing sulfuric acid?
Identify the INCORRECT statement about H2SO4.
The molecular formula of oleum is ____________.
The sulfuric acid obtained by contact process is ____________ % pure.
What is the ratio of volumes of gases involved in the preparation of sulphur trioxide from sulphur dioxide and dioxygen respectively under similar conditions of temperature and pressure?
In the manufacture of sulphuric acid by contact process Tyndall box is used to ____________.
The oxidation number of sulphur in Na2S4O6 is:-
The number of dative bonds in sulphric acid molecule is ______.
What is the catalyst used for the oxidation of SO2 to SO3 in the lead chamber process for the manufacture of sulphuric acid?
