Advertisements
Advertisements
A nitrate, which forms a precipitate with ammonium hydroxide and is also soluble in excess of ______.
Concept: undefined >> undefined
The ratio between the volumes occupied by 4.4 grams of carbon dioxide and 2 grams of hydrogen gas is ______.
Concept: undefined >> undefined
Advertisements
Aqueous lead (II) nitrate can be distinguished from aqueous zinc nitrate by adding any of the following solutions in excess, except:
Concept: undefined >> undefined
Identify the following:
A salt formed by the complete neutralization of an acid by a base.
Concept: undefined >> undefined
Draw the structural diagram for the following compound.
2,2 dibromo butane
Concept: undefined >> undefined
Pratik heated 11.2 grams of element ‘M’ (atomic weight 56) with 4.8 grams of element 'N' (atomic weight 16) to form a compound. Find the empirical formula of the compound obtained by Pratik.
Concept: undefined >> undefined
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
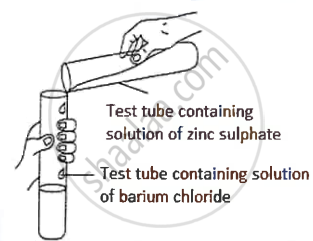
Concept: undefined >> undefined
A student was asked to perform an experiment in the laboratory based on the instructions given:
Observe the picture given below and state one observation for the experiment you would notice on mixing the given solution.
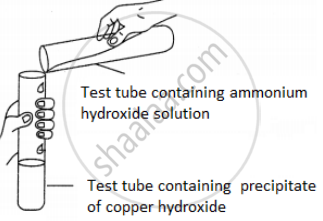
Concept: undefined >> undefined
State giving reasons if calcium nitrate and lead nitrate can be distinguished by adding ammonium hydroxide solution to the salt solution.
Concept: undefined >> undefined
The structures of six organic compounds are shown:
|
A \[\begin{array}{cc} |
B \[\begin{array}{cc} |
C \[\begin{array}{cc} |
|
D \[\begin{array}{cc} |
E \[\begin{array}{cc} |
F \[\begin{array}{cc} |
- Identify two of the compounds that are members of the same homologous series but are not isomers.
- Which two compounds are isomers of each other?
- F can be prepared from D. Give a chemical equation for the reaction.
Concept: undefined >> undefined
You are supplied with five solutions: A, B, C, D and E with pH values as follows: A = 1.8, B = 7, C = 8.5, D = 13, and E = 5
Classify these solutions as neutral, slightly or strongly acidic and slightly or strongly alkaline.
Which solution would be most likely to liberate hydrogen with powdered zinc metal. Give a word equation for each reaction.
Concept: undefined >> undefined
From the formula listed below, choose one, in the following case, corresponding to the salt having the given description:
AgCl, CuCO3, CuSO4.5H2O, KNO3, NaCl, NaHSO4, Pb(NO3)2, ZnCO3, ZnSO4.7H2O.
On treating with concentrated sulphuric acid, this salt changes from blue to white.
Concept: undefined >> undefined
From the formula listed below, choose one, in the following case, corresponding to the salt having the given description:
AgCl, CuCO3, CuSO4.5H2O, KNO3, NaCl, NaHSO4, Pb(NO3)2, ZnCO3, ZnSO4.7H2O.
On heating, this salt changes from green to black.
Concept: undefined >> undefined
From the formula listed below, choose one, in the following case, corresponding to the salt having the given description:
AgCl, CuCO3, CuSO4.5H2O, KNO3, NaCl, NaHSO4, Pb(NO3)2, ZnCO3, ZnSO4.7H2O.
This salt gives nitrogen dioxide on heating.
Concept: undefined >> undefined
The acid which contains four hydrogen atoms ______.
Concept: undefined >> undefined
Match the following in column A with the correct answer from the choices given in column B:
| Column A | Column B |
| 1. Ammonium hydroxide solution | (i) Contains only ions |
| 2. Dilute hydrochloric acid | (ii) Contains only molecules |
| 3. Carbon tetrachloride | (iii) Contains ions and molecules |
Concept: undefined >> undefined
Give a suitable chemical term for:
A salt formed by incomplete neutralisation of an acid by a base.
Concept: undefined >> undefined
Which one of the following will not produce acid with water?
Concept: undefined >> undefined
Select an oxide which dissolves in water forming an acid.
Concept: undefined >> undefined
Draw the structure of Hydronium ion.
Concept: undefined >> undefined
