Advertisements
Advertisements
Write the conditions to maximize the yield of H2SO4 by contact process.
Concept: undefined >> undefined
Why pKa of F-CH2-COOH is lower than that of Cl−CH2−COOH?
Concept: undefined >> undefined
Advertisements
Write the name of vitamin whose deficiency causes bone deformities in children.
Concept: undefined >> undefined
Distinguish between the following : Benzoic acid and methyl benzoate
Concept: undefined >> undefined
What are the different types of emulsions? Give one example of each type.
Concept: undefined >> undefined
Complete the following equations: CaF2+H2SO4 →
Concept: undefined >> undefined
What are the dispersed phase and dispersion medium in milk?
Concept: undefined >> undefined
Complete the following equations: C + conc. H2SO4 →
Concept: undefined >> undefined
Draw the structure of the following:
H2SO4
Concept: undefined >> undefined
How do emulsifires stabilise emulsion? Name two emulsifiers.
Concept: undefined >> undefined
Action of soap is due to emulsification and micelle formation. Comment.
Concept: undefined >> undefined
Give four uses of emulsions.
Concept: undefined >> undefined
Write a short note on the following:
Coupling reaction
Concept: undefined >> undefined
CH3CO2H or CH2FCO2H
Concept: undefined >> undefined
Which acid of the pair shown here would you expect to be stronger?
CH2FCO2H or CH2ClCO2H
Concept: undefined >> undefined
Which acid of the pair shown here would you expect to be stronger?
CH2FCH2CH2CO2H or CH3CHFCH2CO2H
Concept: undefined >> undefined
Which acid of the pair shown here would you expect to be stronger?
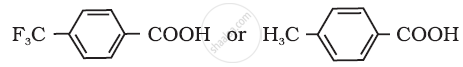
Concept: undefined >> undefined
Arrange the following compounds in increasing order of their property as indicated:
CH3CH2CH(Br)COOH, CH3CH(Br)CH2COOH, (CH3)2CHCOOH, CH3CH2CH2COOH (acid strength)
Concept: undefined >> undefined
Arrange the following compounds in increasing order of their property as indicated:
Benzoic acid, 4-Nitrobenzoic acid, 3, 4-Dinitrobenzoic acid, 4-Methoxybenzoic acid (acid strength)
Concept: undefined >> undefined
