Advertisements
Advertisements
Brine is electrolysed by using inert electrodes. The reaction at anode is ______.
Concept: undefined >> undefined
At temperatures above 1073 K coke can be used to reduce \[\ce{FeO}\] to \[\ce{Fe}\]. How can you justify this reduction with Ellingham diagram?
Concept: undefined >> undefined
Advertisements
Why are sulphide ores converted to oxide before reduction?
Concept: undefined >> undefined
Strong reducing behaviour of \[\ce{H3PO2}\] is due to ______.
Concept: undefined >> undefined
Which of the following statements are correct?
(i) \[\ce{CaF2 + H2SO4 -> CaSO4 + 2HF}\]
(ii) \[\ce{2HI + H2SO4 -> I2 + SO2 + 2H2O}\]
(iii) \[\ce{Cu + 2H2SO4 -> CuSO4 + SO2 + 2H2O}\]
(iv) \[\ce{Nacl + H2SO4 -> NaHSO4 + HCl}\]
Concept: undefined >> undefined
Write a balanced chemical equation for the reaction showing catalytic oxidation of NH3 by atmospheric oxygen.
Concept: undefined >> undefined
Out of \[\ce{H2O}\] and \[\ce{H2S}\], which one has higher bond angle and why?
Concept: undefined >> undefined
Which of the following options are correct for \[\ce{[Fe(CN)6]^{3-}}\] complex?
(i) d2sp3 hybridisation
(ii) sp3d2 hybridisation
(iii) paramagnetic
(iv) diamagnetic
Concept: undefined >> undefined
Magnetic moment of \[\ce{[MnCl4]^{2-}}\] is 5.92 BM. Explain giving reason.
Concept: undefined >> undefined
Explain why \[\ce{[Fe(H2O)6]^{3+}}\] has magnetic moment value of 5.92 BM whereas \[\ce{[Fe(CN)6]^{3-}}\] – has a value of only 1.74 BM.
Concept: undefined >> undefined
Why do compounds having similar geometry have different magnetic moment?
Concept: undefined >> undefined
Assertion: \[\ce{[Fe(CN)6]^{3-}}\] ion shows magnetic moment corresponding to two unpaired electrons.
Reason: Because it has d2sp3 type hybridisation.
Concept: undefined >> undefined
Which of the following compounds will give racemic mixture on nucleophilic substitution by \[\ce{OH-}\] ion?
(a) \[\begin{array}{cc}
\phantom{}\ce{CH3 - CH - Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(b) \[\begin{array}{cc}
\phantom{..}\ce{Br}\\
\phantom{}|\\
\phantom{}\ce{CH3 - C - CH3}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
(c) \[\begin{array}{cc}
\phantom{....}\ce{CH3 - CH - CH2Br}\\
\phantom{}|\\
\phantom{....}\ce{C2H5}\phantom{}
\end{array}\]
Concept: undefined >> undefined
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) | 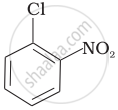 |
| (c) |  |
Concept: undefined >> undefined
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) |  |
| (c) |  |
Concept: undefined >> undefined
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) |  |
| (b) |  |
| (c) |  |
Concept: undefined >> undefined
Arrange the following compounds in increasing order of rate of reaction towards nucleophilic substitution.
| (a) | 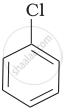 |
| (b) | 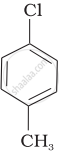 |
| (c) |  |
Concept: undefined >> undefined
Haloarenes are less reactive than haloalkanes and haloalkenes. Explain.
Concept: undefined >> undefined
Allyl chloride is hydrolysed more readily than n-propyl chloride. Why?
Concept: undefined >> undefined
