Advertisements
Advertisements
For an electron in the second orbit of hydrogen, what is the moment of momentum as per the Bohr's model?
Concept: undefined >> undefined
Which of these statements correctly describe the atomic model according to classical electromagnetic theory?
Concept: undefined >> undefined
Advertisements
In Bohr model of hydrogen atom, which of the following is quantised?
Concept: undefined >> undefined
According to Bohr's model of hydrogen atom, an electron can revolve round a proton indefinitely, if its path is ______.
Concept: undefined >> undefined
Explain the terms Inductive and Electromeric effects.
Concept: undefined >> undefined
Hydrogen atom has only one electron, so mutual repulsion between electrons is absent. However, in multielectron atoms mutual repulsion between the electrons is significant. How does this affect the energy of an electron in the orbitals of the same principal quantum number in multielectron atoms?
Concept: undefined >> undefined
When an electric discharge is passed through hydrogen gas, the hydrogen molecules dissociate to produce excited hydrogen atoms. These excited atoms emit electromagnetic radiation of discrete frequencies which can be given by the general formula
`bar(v) = 109677 1/n_1^2 - 1/n_f^2`
What points of Bohr’s model of an atom can be used to arrive at this formula? Based on these points derive the above formula giving description of each step and each term.
Concept: undefined >> undefined
Calculate the energy and frequency of the radiation emitted when an electron jumps from n = 3 to n = 2 in a hydrogen atom.
Concept: undefined >> undefined
Why was a change in the Bohr Model of atom required? Due to which important development (s), concept of movement of an electron in an orbit was replaced by, the concept of probability of finding electron in an orbital? What is the name given to the changed model of atom?
Concept: undefined >> undefined
Ionisation enthalpies of elements of second period are given below: Ionisation enthalpy/ k cal mol–1:
520, 899, 801, 1086, 1402, 1314, 1681, 2080.

Match the correct enthalpy with the elements and complete the graph given in figure. Also write symbols of elements with their atomic number
Concept: undefined >> undefined
First member of each group of representative elements (i.e., s and p-block elements) shows anomalous behaviour. Illustrate with two examples.
Concept: undefined >> undefined
A plot of volume (V) versus temperature (T) for a gas at constant pressure is a straight line passing through the origin. The plots at different values of pressure are shown in Figure. Which of the following order of pressure is correct for this gas?
Concept: undefined >> undefined
Under which of the following two conditions applied together, a gas deviates most from the ideal behaviour?
(i) Low pressure
(ii) High pressure
(iii) Low temperature
(iv) High temperature
Concept: undefined >> undefined
If 1 gram of each of the following gases are taken at STP, which of the gases will occupy (a) greatest volume and (b) smallest volume?
\[\ce{CO, H2O, CH4 , NO}\]
Concept: undefined >> undefined
Value of universal gas constant (R) is same for all gases. What is its physical significance?
Concept: undefined >> undefined
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. What is the value of Z for an ideal gas?
Concept: undefined >> undefined
Compressibility factor, Z, of a gas is given as Z = `(pV)/(nRT)`. For real gas what will be the effect on value of Z above Boyle’s temperature?
Concept: undefined >> undefined
Pressure versus volume graph for a real gas and an ideal gas are shown in figure. Answer the following questions on the basis of this graph.
(i) Interpret the behaviour of real gas with respect to ideal gas at low pressure.
(ii) Interpret the behaviour of real gas with respect to ideal gas at high pressure.
(iii) Mark the pressure and volume by drawing a line at the point where real gas behaves as an ideal gas.
Concept: undefined >> undefined
Match the following graphs of ideal gas with their co-ordinates:
| Graphical representation | x and y co-ordinates |
(i) 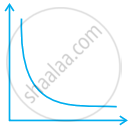 |
(a) pV vs. V |
(ii) 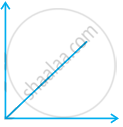 |
(b) p vs. V |
(iii) 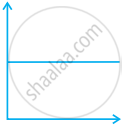 |
(c) p vs. `1/V` |
Concept: undefined >> undefined
Assertion (A): At constant temperature, pV vs V plot for real gases is not a straight line.
Reason (R): At high pressure all gases have \[\ce{Z}\] > 1 but at intermediate pressure most gases have \[\ce{Z}\] < 1.
Concept: undefined >> undefined
