Advertisements
Advertisements
Question
Write the molecular formula of first two members of homologous series having functional group – OH
Advertisements
Solution
The general formula for the homologous series of functional group −OH is CnH2n+1OH.
The two consecutive members of this series are:
a. CH3OH(Methanol)
b. CH3CH2OH (Ethanol)
where n is 1 and 2 respectively
The molecular formula for both of them are CH4O and C2H6O.
APPEARS IN
RELATED QUESTIONS
Write the molecular formula of two consecutive members of homologous series of aldehydes. State which part of these compounds determines their
- physical and
- chemical properties
The following vegetables are kept in a basket :
Potato, Tomato, Radish, Brinjal, Carrot, Bottle-gourd
Which two of these vegetables correctly represent the homologous structures?
(A) Carrot and Tomato
(B) Potato and Brinjal
(C) Radish and Carrot
(D) Radish and Bottle-gourd
Give the structural formula for 1, 2-dichloroethane
The molecular formula of an organic compound is C18H36. Name its homologous series.
Propane and ethane are ______.
 .
.
Distinguish between homologous organs and analogous organs. In which category would you place wings of a bird and wings of a bat? Justify your answer giving a suitable reason.
Observe the structural formula and answer the following questions.
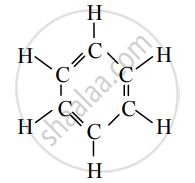
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Which of the following pairs can be the successive members of a homologous series?
Name the following organic compound:
The first homologue whose general formula is CnH2n.
