Advertisements
Advertisements
प्रश्न
Write the molecular formula of first two members of homologous series having functional group – OH
Advertisements
उत्तर
The general formula for the homologous series of functional group −OH is CnH2n+1OH.
The two consecutive members of this series are:
a. CH3OH(Methanol)
b. CH3CH2OH (Ethanol)
where n is 1 and 2 respectively
The molecular formula for both of them are CH4O and C2H6O.
APPEARS IN
संबंधित प्रश्न
Write the name and formula of the 2nd member of homologous series having general formula CnH2n + 2.
Give the molecular formula of one homologue of the following:
C3H6
An organic compound having the molecular formula C3H6O can exist in the form of two isomers A and B having different functional groups. The isomer A is a liquid which is used as a solvent for nail polish. The isomer B is also a liquid. An aqueous solution of one of the lower homologues of B is used for preserving biological specimens in the laboratory
(a) What is compound A?
(b) Write the electron-dot structure of A.
(c) What is compound B?
(d) Write the electron-dot structure of B.
(e) Name the lower homologue of compound B which is used in preserving biological specimens.
 .
.
Haloalkanes react with alkalies to produce alcohol. Give the equation for the preparation of the second member of the homologous series of alcohol. State under what condition the reaction occurs.
While going in an increasing order there is a rise in the molecular mass of the consecutive members of the homologous series by _______.
Saturated hydrocarbon : Single bond : : Unsaturated hydrocarbon : _______
Complete the following table for the homologous series of alkanes.
| Name | Molecular formula | Condensed structural formula | Number of carbon atom | Number of -CH2- units | Boiling point °C |
| Methane | CH4 | CH4 | 1 | 1 | -162 |
| Ethane | C2H6 | CH3–CH3 | 2 | 2 | -88.5 |
| Propane | C3H8 | CH3–CH2–CH3 | 3 | 3 | -42 |
| Butane | C4H10 | CH3–CH2–CH2–CH3 | ______ | ______ | 0 |
| Pentane | C5H12 | CH3–CH2–CH2–CH2–CH3 | ______ | ______ | 36 |
| Hexane | C6H14 | CH3–CH2–CH2–CH2–CH2–CH3 | ______ | ______ | 69 |
Observe the structural formula and answer the following questions.
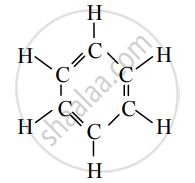
- Write the name of the given hydrocarbon.
- The given hydrocarbon is included in which type of hydrocarbon?
- What is the kind of compounds with the above characteristic structure called?
Which of the following does not belong to the same homologous series?
