Advertisements
Advertisements
Questions
Write the electron-dot structure for the following molecule.
Cl2
Atomic number of chlorine is 17.
Draw the electron dot structure of a chlorine molecule.
Advertisements
Solution
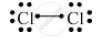
APPEARS IN
RELATED QUESTIONS
Buckminsterfullerene is spherical molecule in which 60 carbon atoms are arranged in interlocking hexagonal and pentagonal rings of carbon atoms.
How many pentagons of carbon atoms are present in one molecule of buckminsterfullerene?
What type of bond exist in CCl4?
Why does carbon form compounds mainly by covalent bonding?
What is buckminsterfullerene?
will CS2 conduct electricity or not?
give reason for your choice.
Give the formula of the compound that would be formed by the combination of the following pair of elements:
K and H
Why is graphite a good conductor of electricity but diamond is a non-conductor of electricity?
One of the following compounds is not ionic in nature. This compound is:
(a) Lithium chloride
(b) Ammonium chloride
(c) Calcium chloride
(d) Carbon tetrachloride
Give two example in following case:
Gaseous polar compounds
Distinguish between ionic and covalent compounds under the following properties:
(i) Strength of forces between constituent elements
(ii) Solubility of compounds in water
(iii) Electrical conduction in substances
Compare the compounds carbon tetrachloride and sodium chloride with regard to solubility in water and electrical conductivity.
Electrons are getting added to an element Y:
which electrode will Y migrate to during the process of electrolysis?
Draw the electron dot diagram and structure of magnesium chloride.
Name gas with a rotten egg smell.
Fill in the blank and rewrite the completed statement:
Covalent compounds are generally soluble in _________ solvents.
Considering MgCl2 as ionic compound and CH4 as covalent compound give any two differences between these two compounds.
Discuss in brief about the properties of coordinate covalent compounds.
Consider the coordination compound, K2[Cu(CN)4]. A coordinate covalent bond exists between:
In covalent bond formation, the sharing of ______ electrons takes place in their outermost shell.
