Advertisements
Advertisements
प्रश्न
Write the electron-dot structure for the following molecule.
Cl2
Atomic number of chlorine is 17.
Draw the electron dot structure of a chlorine molecule.
Advertisements
उत्तर
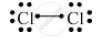
APPEARS IN
संबंधित प्रश्न
Elements forming ionic compounds attain noble gas configuration by either gaining or losing electrons from their outermost shells. Give reason to explain why carbon cannot attain noble gas configuration in this manner to form its compounds.
What type of chemical bond is formed between carbon and bromine?
State one major difference between covalent and ionic bonds and give one example each of covalent and ionic compounds.
What type of bonds are present in CH4 molecule? Draw their electron-dot structures.
Fill in the blank in the following sentence:
In forming N2 molecule, .............. electrons are shared by each atom of nitrogen.
What is a covalent bond?
Draw the electron-dot structure of HCl compound and state the type of bonding.
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of oxygen, O2, is formed from two oxygen atoms. What name is given to this type of bonding? (At. No. of oxygen = 8)
Give two example in following case:
Solid covalent compounds
Give examples for the following:
Two liquid non polar compounds.
(a) Compound X consists of molecules.
Choose the letter corresponding to the correct answer from the choices (a), (b), (c) and (d) given below
The type of bonding in X will be
What are the term defined below?
A bond formed by share pair of electrons, each bonding atom contributing one electron to the pair.
Draw the electron dot structure of covalent compound methane (non polar) and HCL (polar) and give two difference between them.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
The number of electrons in the valence shell of a carbon atom is 4.
Write an Explanation.
Alkyne
When ethyl alcohol and acetic acid are mixed, the resulting ester has a chemical formula ______.
Which of the following is the formula of Butanoic acid?
Statement (A): Covalent compounds are bad conductors of electricity.
Reason (B): Covalent compounds contain charged particles (ions)
