Advertisements
Advertisements
प्रश्न
Write the electron-dot structure for the following molecule.
Cl2
Atomic number of chlorine is 17.
Draw the electron dot structure of a chlorine molecule.
Advertisements
उत्तर
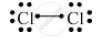
APPEARS IN
संबंधित प्रश्न
State whether the following statement is true or false:
Diamond and graphite are the covalent compounds of carbon element (C)
What type of bonding would you expect between Carbon and Chlorine?
Using electron-dot diagrams which show only the outermost shell electrons, show how a molecule of nitrogen, N2, is formed from two nitrogen atoms. What name is given to this type of bonding? (Atomic number of nitrogen is 7)
The number of covalent bonds in pentane (molecular formula C5H12) is:
The organic compound prepared by Wohler from an inorganic compound called ammonium cyanate was:
(a) glucose
(b) urea
(c) uric acid
(d) vinegar
The electronic configurations of three elements X, Y and Z are as follows:
| X | 2, 4 |
| Y | 2, 7 |
| Z | 2, 1 |
(a) Which two elements will combine to form an ionic compound?
(b) Which two elements will react to form a covalent compound?
Give reasons for your choice.
Draw an electron dot structure of the following molecule. (Without showing the circle) :
Methane
Explain the following term with example.
Hetero atom in a carbon compound
Explain the structure of Hydronium ion.
Explain the structure of Ammonium ion.
Choose the correct answer from the options given below
Condition favorable for formation of a covalent bond is
State the type of bonding in the following molecule.
Water
Potassium (Atomic No. 19) and chlorine (Atomic No. 17) react to form a compound. On the basis of electronic concept, explain
(i) oxidation
(ii) reduction
(iii)oxidising agent
(iv)reducing agent
Covalent bonds can be single, double or triple covalent bonds. How many electrons are shared in each? Give an example of each type.
Name the types of Hydrocarbons.
From the following hydrocarbon _______ is the cyclic hydrocarbon.
Write a short note.
Aromatic hydrocarbons
When ethyl alcohol and acetic acid are mixed, the resulting ester has a chemical formula ______.
It is observed that covalent compounds are had conductors of electricity. Give reason.
Non-polar covalent compounds are ______ conductors of heat and electricity.
