Advertisements
Advertisements
Question
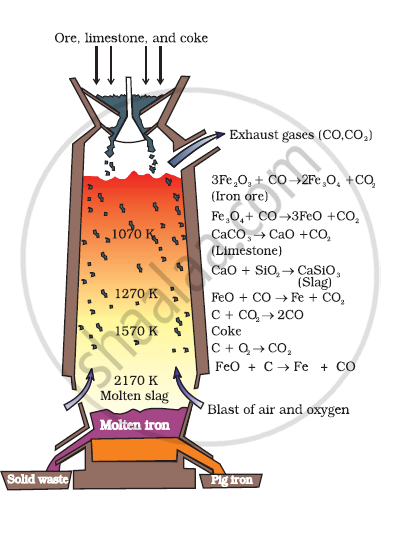
Write down the reactions taking place in different zones in the blast furnace during the extraction of iron.
Advertisements
Solution
During the extraction of iron, the reduction of iron oxides takes place in the blast furnace. In this process, hot air is blown from the bottom of the furnace, and coke is burnt to raise the temperature up to 2200 K in the lower portion itself. The temperature is lower in the upper part. Thus, it is the lower part where the reduction of iron oxides (Fe2O3 and Fe3O4) takes place.
The reactions taking place in the lower temperature range (500 − 800 K) in the blast furnace are:
\[\ce{3Fe2O3 + CO -> 2Fe3O4 + CO2}\]
\[\ce{Fe3O4 + 4CO -> 3Fe + 4CO2}\]
\[\ce{Fe2O3 + CO -> 2FeO + CO2}\]
The reactions taking place in the higher temperature range (900-1500 K) in the blast furnace are:
\[\ce{C + CO2 -> 2CO}\]
\[\ce{FeO + CO -> Fe + CO2}\]
The silicate impurity of the ore is removed as slag by calcium oxide (CaO), which is formed by the decomposition of limestone (CaCO3)
\[\ce{CaCO3 -> CaO + CO2}\]
\[\ce{CaO + SiO2 -> \underset{(Slag)}{\underset{Calcium silicate}{CaSiO3}}}\]
RELATED QUESTIONS
Is it true that under certain conditions, Mg can reduce SiO2 and Si can reduce MgO? What are those conditions?
Why is the extraction of copper from pyrites more difficult than that from its oxide ore through reduction?
Write chemical reactions taking place in the extraction of zinc from zinc blende.
State the role of silica in the metallurgy of copper.
The impurity that is added externally to remove the impurity already present in the ore is known as ____________.
In the metallurgy of aluminium ______.
Choose the correct option of temperature at which carbon reduces \[\ce{FeO}\] to iron and produces \[\ce{CO}\].
For the reduction of \[\ce{FeO}\] at the temperature corresponding to point D, which of the following statements is correct?
Wrought iron is the purest form of iron. Write a reaction used for the preparation of wrought iron from cast iron. How can the impurities of sulphur, silicon and phosphorus be removed from cast iron?
How is copper extracted from low grade copper ores?
Write down the reactions taking place in Blast furnace related to the metallurgy of iron in the temperature range 500-800 K.
Explain the following:
Generally sulphide ores are converted into oxides before reduction.
Explain the following:
Silica is added to the sulphide ore of copper in the reverberatory furnace.
Explain the following:
Carbon and hydrogen are not used as reducing agents at high temperatures.
A cuprous ore among the following is:-
\[\ce{Au + CN^- + H2O + O2 -> [Au(CN)2]^- + OH^-}\]
The number of CN− ions involved in the balanced equation is ______.
