Advertisements
Advertisements
Question
With the help of a labeled diagram, describe a simple experiment to show the presence of oxygen and nitrogen in air using a piece of white phosphorus.
Advertisements
Solution
Activity to show the presence of oxygen and nitrogen in the air using a piece of white phosphorous
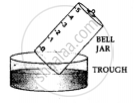
1. A trough is taken and filled with water
2. Over it is placed a bell-jar marked with five equal parts.
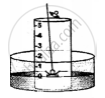
3. A crucible containing white phosphorus is placed on a cork
4. The cork is made to float on the water.
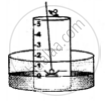
5. A heated iron rod is them taken.
6. The phosphorus is ignited with the heated rod.
Observations:
Dense white fumes of phosphorus pentoxide (P2O5) are formed when the phosphorus (P) burns in the active component of air (oxygen).
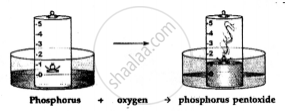
The rise in the level of water in the bell-jar is by 1/5
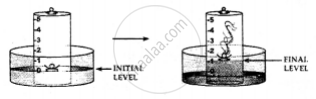
Conclusion:
|
Oxygen (active compound) |
1/5 (approx volume) | Supports combustion | Used up in burning |
|
Nitrogen (In active component bell - jar) |
4/5 (approx volume) | Does not support combustion | Remains behind in the combustion |
The active component of air i.e. oxygen (1/5 of air) is used up in burning. The remaining inactive component of air i.e. nitrogen (4/ 5 of air) is not used up in burning.
RELATED QUESTIONS
Fill in the blank :
________ is the most abundant inert gas present in air.
Statement given below is incorrect. Write the correct statement :
Potassium nitrate on strong heating decomposes into potassium nitrite and nitrogen gas.
Fill in the blanks
Plants and animals maintain the balance of carbon ___________and _________ in the air.
Fill in the blank
Green plants need ________________to prepare food.
Write ‘true’ or ‘false for the following;
Carbon dioxide is given out during photosynthesis
Name the main component of air.
Air is made up of oxygen only.
Arrange the following statements in correct sequence
- Plants manufacture food by a process called photosynthesis.
- Plants require energy for their growth.
- Plants take in oxygen and release carbon dioxide just as animals.
- Plants take carbon dioxide from the atmosphere, use chlorophyll in the presence of sunlight and prepare food.
- Such oxygen is available to animals and human beings for breathing.
- During this process, oxygen is released by plants.
Tri oxygen molecule is known as ______.
On a Sunday morning Paheli’s friend visited her home. She wanted to see some flowering plants in the nearby garden. Both of them went to the garden. While returning from the garden they also observed some flowering plants on the road side. But to their surprise they found that the leaves and flowers of these roadside plants were comparatively very dull. Can you help them to know why?
