Advertisements
Advertisements
प्रश्न
With the help of a labeled diagram, describe a simple experiment to show the presence of oxygen and nitrogen in air using a piece of white phosphorus.
Advertisements
उत्तर
Activity to show the presence of oxygen and nitrogen in the air using a piece of white phosphorous
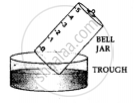
1. A trough is taken and filled with water
2. Over it is placed a bell-jar marked with five equal parts.
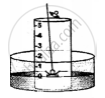
3. A crucible containing white phosphorus is placed on a cork
4. The cork is made to float on the water.
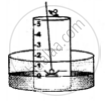
5. A heated iron rod is them taken.
6. The phosphorus is ignited with the heated rod.
Observations:
Dense white fumes of phosphorus pentoxide (P2O5) are formed when the phosphorus (P) burns in the active component of air (oxygen).
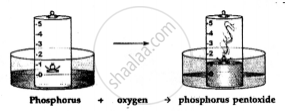
The rise in the level of water in the bell-jar is by 1/5
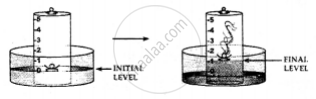
Conclusion:
|
Oxygen (active compound) |
1/5 (approx volume) | Supports combustion | Used up in burning |
|
Nitrogen (In active component bell - jar) |
4/5 (approx volume) | Does not support combustion | Remains behind in the combustion |
The active component of air i.e. oxygen (1/5 of air) is used up in burning. The remaining inactive component of air i.e. nitrogen (4/ 5 of air) is not used up in burning.
संबंधित प्रश्न
Describe a simple experiment to show that l/5th of air is oxygen.
Briefly state the importance of Oxygen.
Give reason:
When water is heated, we see bubbles rising up.
Name the following:
The component of air which is present more in industrial areas.
Explain why during an incident of fire, one is advised to wrap a woollen blanket over a burning object.
Match the following
| 1. | Moving Air | Photosynthesis |
| 2. | Layer in which we live | Troposphere |
| 3. | Stratosphere | Wind |
| 4. | Oxygen | Ozone layer |
| 5. | carbon-di-oxide | Combustion |
Nitrogen is _______ than air.
The main component of air is oxygen.
The amount of water vapour in the air is more in windy places than in other areas.
Garima observed that when she left her tightly capped bottle full of water in the open sunlight, tiny bubbles were formed all around inside the bottle. Help Garima to know why it so happened?
