Advertisements
Advertisements
प्रश्न
With the help of a labeled diagram, describe a simple experiment to show the presence of oxygen and nitrogen in air using a piece of white phosphorus.
Advertisements
उत्तर
Activity to show the presence of oxygen and nitrogen in the air using a piece of white phosphorous
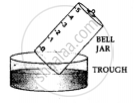
1. A trough is taken and filled with water
2. Over it is placed a bell-jar marked with five equal parts.
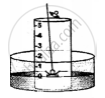
3. A crucible containing white phosphorus is placed on a cork
4. The cork is made to float on the water.
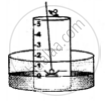
5. A heated iron rod is them taken.
6. The phosphorus is ignited with the heated rod.
Observations:
Dense white fumes of phosphorus pentoxide (P2O5) are formed when the phosphorus (P) burns in the active component of air (oxygen).
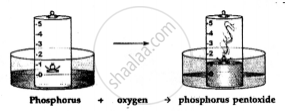
The rise in the level of water in the bell-jar is by 1/5
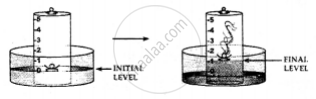
Conclusion:
|
Oxygen (active compound) |
1/5 (approx volume) | Supports combustion | Used up in burning |
|
Nitrogen (In active component bell - jar) |
4/5 (approx volume) | Does not support combustion | Remains behind in the combustion |
The active component of air i.e. oxygen (1/5 of air) is used up in burning. The remaining inactive component of air i.e. nitrogen (4/ 5 of air) is not used up in burning.
संबंधित प्रश्न
Suggest five measures to prevent air pollution.
Statement given below is incorrect. Write the correct statement :
Carbon dioxide gas allows the earth to radiate out heat rapidly.
Name and briefly explain four ways by which pollution of air caused by humans is minimised.
What is the significance of water vapour in air?
Give reason:
Mountaineers and divers carry oxygen cylinders with them.
State the utility of oxygen for respiration in plants
Complete the statement given below by filling in the blank with the correct word from the words given:
The product formed which is common to both combustion and respiration is ______.
What is the source of carbon dioxide in air?
Match the following.
| Nitrogen | Respiration in living animals |
| Oxygen | Fertilizer |
| Carbon dioxide | Refrigerator |
| Dry ice | Fire extinguisher |
Explain carbon dioxide and its uses.
