Advertisements
Advertisements
प्रश्न
You are given a test tube with two outlets and a bottle of lime water. Using same, how would you demonstrate experimentally the presence of carbon dioxide in air.
Advertisements
उत्तर
Procedure:
- Take lime water in a test tube with two outlets ‘A’ and ‘B’ as shown above.
- Air is sucked slowly through the outlet ‘B’.
- Air moves in through end ‘A’ and passes through the lime water
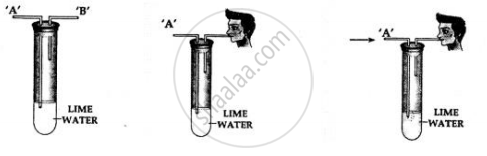
Observation:
The line water in the test tube turns milky.
Conclusion:
Air which is sucked in from and ‘A’ contains carbon dioxide which turns the lime water milky. Hence, air contains – carbon dioxide.
संबंधित प्रश्न
Complete and balance the following chemical equation.
CaO + H2O → ____________
Tick (√) the most appropriate answer.
Symbiotic bacteria is not present in the nodules of :
Classify Carbon monoxide. Also give a balanced equation to prepare it.
State the full form of LPG and CNG? How are the two different in their composition?
Compare respiration and combustion-both involving oxygen of the air.
Can you guess why fire extinguishers throw a stream of carbon-di-oxide while putting - off the fire?
How will you show that air is dissolved in water?
What was the old name of nitrogen?
The second major component of air is oxygen.
Write just a few sentences for an imaginary situation if any of the following gases disappear from the atmosphere.
Oxygen
