Advertisements
Advertisements
Question
Which of the following solutions have the same concentration?
(i) \[\ce{20 g of NaOH in 200 mL of solution}\]
(ii) \[\ce{0.5 mol of KCl in 200 mL of solution}\]
(iii) \[\ce{40 g of NaOH in 100 mL of solution}\]
(iv) \[\ce{20 g of KOH in 200 mL of solution}\]
Advertisements
Solution
(i) \[\ce{20 g of NaOH in 200 mL of solution}\]
(ii) \[\ce{0.5 mol of KCl in 200 mL of solution}\]
Explanation:
(i) The number of moles is given by the following formula,
Moles = `"Mass"/"Molar mass"` ......(1)
So, the number of moles of \[\ce{NaOH}\] is calculated, by using equation (1) as follows,
Moles of NaOH = `(20 g)/((40 g)/(mol)` = 0.5 mol
The molarity (M) is given by the formula:
M = `n/V_((L))`
On substituting the values in the above equation:
MNaCH = `(0.5 mol xx 1000)/(200 L)` = 2.5 mol L–1
(ii) The molarity is given by the formula:
M = `n/V_((L))`
On substituting the values in the above equation:
MKCl = `(0.5 mol xx 1000)/(200 L)` = 2.5 mol L–1
APPEARS IN
RELATED QUESTIONS
Explain the term Saturated solution giving examples.
Dinitrogen and dihydrogen react with each other to produce ammonia according to the following chemical equation:
\[\ce{N2 (g) + 3H2 (g) → 2NH3 (g)}\]
(i) Calculate the mass of ammonia produced if 2.00 × 103 g dinitrogen reacts with 1.00 × 103 g of dihydrogen.
(ii) Will any of the two reactants remain unreacted?
(iii) If yes, which one and what would be its mass?
Calculate the molarity of a solution of ethanol in water in which the mole fraction of ethanol is 0.040 (assume the density of water to be one).
Why does the molarity of a solution depend upon temperature?
Solve the following problem:
Write the following number in ordinary decimal form:
5.16 × 104
Solve the following problem:
Write the following number in ordinary decimal form:
14.3 × 10−2
Perform each of the following calculations. Round off your answers to three digits.
(3.26104) (1.54106)
A 1.000 mL sample of acetone, a common solvent used as a paint remover, was placed in a small bottle whose mass was known to be 38.0015 g.
The following values were obtained when the acetone-filled bottle was weighed: 38.7798 g, 38.7795 g and 38.7801 g. How would you characterise the precision and accuracy of these measurements if the actual mass of the acetone was 0.7791 g?
Solve the following problem:
Your laboratory partner was given the task of measuring the length of a box (approx 5 in) as accurately as possible, using a metre stick graduated in milimeters. He supplied you with the following measurements:
12.65 cm, 12.6 cm, 12.65 cm, 12.655 cm, 126.55 mm, 12 cm.
Give your reason for rejecting each of the others.
What are the favourable qualities given to gold when it is alloyed with copper or silver for the purpose of making ornaments?
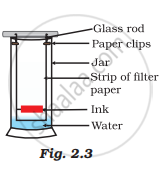
A child wanted to separate the mixture of dyes constituting a sample of ink. He marked a line by the ink on the filter paper and placed the filter paper in a glass containing water as shown in Fig.2.3. The filter paper was removed when the water moved near the top of the filter paper.
(i) What would you expect to see, if the ink contains three different coloured components?
(ii) Name the technique used by the child.
(iii) Suggest one more application of this technique.
What will be the molality of the solution containing 18.25 g of \[\ce{HCl}\] gas in 500 g of water?
Sulphuric acid reacts with sodium hydroxide as follows:
\[\ce{H2SO4 + 2NaOH -> Na2SO4 + 2H2O}\]
When 1 L of 0.1 M sulphuric acid solution is allowed to react with 1 L of 0.1 M sodium hydroxide solution, the amount of sodium sulphate formed and its molarity in the solution obtained is:
(i) 0.1 mol L–1
(ii) 7.10 g
(iii) 0.025 mol L–1
(iv) 3.55 g
The molarity of pure water is ______.
What quantity (in mL) of a 45% acid solution of a monoprotic strong acid must be mixed with a 20% solution of the same acid to produce 800 mL of a 29.875% acid solution?
Molarity of liquid HCl will be if the density of the solution is 1.17 g/cc.
Find the molality of solution if boiling point increases by 1.75 K and molal elevation constant of solvent is 5K kg mol-1.
