Advertisements
Advertisements
Question
Water is cooled from 4 °C to 0 °C. It will :
Options
contract
expand
first contract, then expand
first expand, then contract
Advertisements
Solution
Water shows anomalous behavior between 0 °C and 4 °C. Hence, when it is cooled it expands.
APPEARS IN
RELATED QUESTIONS
On what basis and how will you determine whether air is saturated with vapour or not?
A 50 kg man is running at a speed of 18 km h−1. If all the kinetic energy of the man can be used to increase the temperature of water from 20°C to 30°C, how much water can be heated with this energy?
Draw a graph to show the variation in density of water with temperature in the range from 0°C to 10°C.
What are hot spots? How can you extract energy from a hot spot, if it does not come in contact with underground water?
What will be the approximate temperature of water in the lake shown in the following diagram?

(i) At X, and
(ii) At Y?
When the temperature of water decreases below 4 °C it’s volume _______.
Write scientific reason.
Fish can survive even in frozen ponds in cold regions.
Write scientific reason.
Placing a plastic bottle filled with water in the freezing compartment in the freezer can cause the bottle to explode.
Observe the following diagram and write the answers to the questions given below.
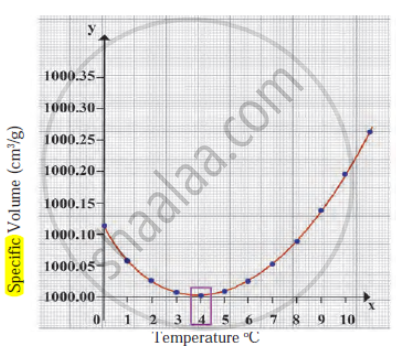
- Which process does the graph represent?
- What is the range of temperature responsible for the behaviour?
