Advertisements
Advertisements
Question
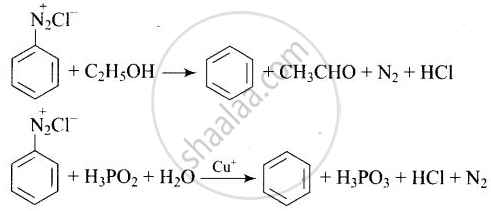
The reagents that can be used to convert benzenediazonium chloride to benzene are:
(i) \[\ce{SnCl2/HCl}\]
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
(iv) \[\ce{LiAlH4}\]
Advertisements
Solution
(ii) \[\ce{CH3CH2OH}\]
(iii) \[\ce{H3PO2}\]
Explanation:
APPEARS IN
RELATED QUESTIONS
Write a short note on Hoffmann bromamide degradation.
Accomplish the following conversion:
Benzamide to toluene
Why cannot aromatic primary amines be prepared by Gabriel phthalimide synthesis?
Identify 'A' and 'B' in the following reaction and rewrite the complete reaction :

Give the structures of A, B and C in the following reactions :

Acetamide on reduction using Na/C2H5OH gives ____________.
Alkyl cyanides on reduction by sodium and ethanol give primary amines. This reaction is called as ____________.
For producing amines, the reaction of nitro compounds with iron scrap is preferred because:
Given below are two statements labelled as Assertion (A) and Reason (R).
Assertion (A): Alkyl halides are insoluble in water.
Reason (R): Alkyl halides have halogen attached to sp3 hybrid carbon.
Select the most appropriate answer from the options given below:
Which of the following amines can be prepared by Gabriel synthesis.
(i) Isobutyl amine
(ii) 2-Phenylethylamine
(iii) N-methylbenzylamine
(iv) Aniline
Which of the following reactions are correct?
(i)

(ii)

(iii)

(iv)

What is the best reagent to convert nitrile to primary amine?
Identify A and B in the following reaction.

The Gabriels' phthalimide synthesis is used in the synthesis of
In the given reaction what is the X?
\[\begin{array}{cc}
\ce{O}\phantom{.......................}\\
||\phantom{.......................}\\
\phantom{}\ce{R - C - OH <-[H3O] Χ ->[H] RCH2NH2}
\end{array}\]
Which of the following CANNOT be prepared by ammonolysis of alkyl halide?
Give reasons for the following:
Ammonolysis of alkyl halides is not a good method to prepare pure primary amines.
Write short note on the following:
Ammonolysis
