Advertisements
Advertisements
Question
The experimental data for decomposition of N2O5 \[\ce{2N2O5 -> 4NO2 + O2}\] in gas phase at 318 K are given below:
| t/s | 0 | 400 | 800 | 1200 | 1600 | 2000 | 2400 | 2800 | 3200 |
| 102 × [N2O5]/mol L−1 | 1.63 | 1.36 | 1.14 | 0.93 | 0.78 | 0.64 | 0.53 | 0.43 | 0.35 |
- Plot [N2O5] against t.
- Find the half-life period for the reaction.
- Draw a graph between log [N2O5] and t.
- What is the rate law?
- Calculate the rate constant.
- Calculate the half-life period from k and compare it with (ii).
Advertisements
Solution
| t/s | [N2O5] × 102/mol L−1 | log [N2O5] |
| 0 | 1.63 | −1.79 |
| 400 | 1.36 | −1.87 |
| 800 | 1.14 | −1.94 |
| 1200 | 0.93 | −2.03 |
| 1600 | 0.78 | −2.11 |
| 2000 | 0.64 | −2.19 |
| 2400 | 0.53 | −2.28 |
| 2800 | 0.43 | −2.37 |
| 3200 | 0.35 | −2.46 |
i.
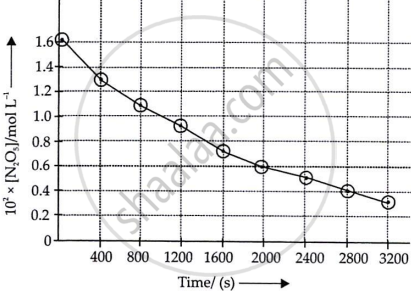
ii. Initial concentration of N2O5 = 1.63 × 10−2 M
Half of this concentration = 0.815 × 10−2 M
The time corresponding to M concentration = 1440 s
Hence, t1/2 = 1440 s
iii.
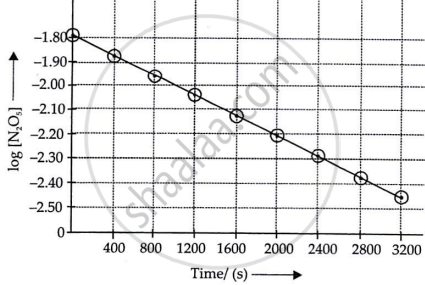
iv. The given reaction is of the first order as the plot, log [N2O5] v/s t, is a straight line. Hence, it is a reaction of first order, i.e., the rate law is:
Rate = k[N2O5]
v. For first order reaction,
log R = `- k/(2.303 t) + log R_0`
Therefore, the slope of the graph drawn between log R and t will be `(-k)/2.303`.
∴ The slope of the line = `-k/2.303`
= `(y_2 - y_1)/(t_2 - t_1)`
= `(-2.46 - (-1.79))/(3200 - 0)`
= `-0.67/3200`
∴ k = 4.82 × 10−4 s−1
vi. Half-life is given by,
t1/2 = `0.639/k`
= `0.693/(4.82 xx 10^-4)`
= 1.438 × 103 s
= 1438 s
This value, 1438 s, is very close to the value that was obtained from the graph.
APPEARS IN
RELATED QUESTIONS
For a first order reaction, show that time required for 99% completion is twice the time required for the completion of 90% of reaction.
A first order reaction takes 40 minutes for 30% decomposition. Calculate t1/2 for this reaction. (Given log 1.428 = 0.1548)
A first order reaction takes 30 minutes for 50% completion. Calculate the time required for 90% completion of this reaction.
(log 2 = 0.3010)
During nuclear explosion, one of the products is 90Sr with half-life of 28.1 years. If 1μg of 90Sr was absorbed in the bones of a newly born baby instead of calcium, how much of it will remain after 10 years and 60 years if it is not lost metabolically.
The unit of rate constant for zero order reaction is _______.
(A) t–1
(B) mol dm–3 t–1
(C) mol–1 dm3 t–1
(D) mol–2 dm6 t–1
A first order reaction takes 10 minutes for 25% decomposition. Calculate t1/2 for the reaction.
(Given : log 2 = 0.3010, log 3 = 0.4771, log 4 = 0.6021)
The half life period of a first order reaction is 6. 0 h . Calculate the rate constant
In a first-order reaction A → product, 80% of the given sample of compound decomposes in 40 min. What is the half-life period of the reaction?
After 2 hours, a radioactive substance becomes `(1/16)^"th"` of original amount. Then the half life ( in min) is
Half life (t1/2) and completion time (T) of the zero order reaction are- (K = 0.001 mol/litre/sec and a = 1 M.)
Observe the graph shown in figure and answer the following questions:

Write the relationship between k and t1/2 (half-life period)
A sample of U238 (half-life = 4.5 × 109 years) ore is found to contain 23.8 g of U238 and 20.6 g of Pb206. The age of the ore is ______ × 109 years.
The half-life of cobalt 60 is 5.26 years. The percentage activity remaining after 4 years is ______%.
For the given first order reaction A → B the half life of the reaction is 0.3010 min. The ratio of the initial concentration of reactant to the concentration of reactant at time 2.0 min will be equal to ______. (Nearest integer)
A reaction has a half-life of 1 min. The time required for 99.9% completion of the reaction is ______ min.
(Round off to the nearest integer).
[Use: In 2 = 0.69; In 10 = 2.3]
Assertion (A): The half-life of a reaction is the time in which the concentration of the reactant is reduced to one-half of its initial concentration.
Reason (R): In first-order kinetics, when the concentration of reactant is doubled, its half-life is doubled.
Calculate the half-life of a first order reaction from the rate constant given below:
2 min−1
A first order reaction takes 40 min for 30% decomposition. Calculate t1/2.
