Advertisements
Advertisements
Question
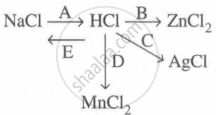
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Advertisements
Solution
A: \[\ce{NaCl + H2SO4 ->[< 200^\circ C] NaHSO4 + HCl ^}\]
B: \[\ce{Zn + 2HCl -> ZnCl2 + H2}\]
C: \[\ce{AgNO3 + HCl -> AgCl + HNO3}\]
D: \[\ce{MnO2 + 4HCl ->[\Delta] MnCl2 + 2H2O + Cl2 ^}\]
E: \[\ce{HCl + NaOH -> NaCl + H2O}\]
APPEARS IN
RELATED QUESTIONS
Name the experiment which demonstrates that hydrogen chloride is soluble in water.
Name the gas evolved when dilute hydrochloric acid is added to: Lead (II) sulphide
Give a balanced equation when dilute hydrochloric acid is added to : Potassium bisulphite
How will you identify?
Chloride ion
A solution of hydrogen chloride in water is prepared. The following substances are added to separate portions of the solution:
| S. No. | Substances added | Gas evolved | Odour |
| 1. | Calcium carbonate | _________ | _________ |
| 2. | Magnesium ribbon | _________ | _________ |
| 3. | Manganese (IV) oxide with heating | _________ | _________ |
| 4. | Sodium sulphide | _________ | _________ |
Complete and balance the following reaction, state whether dilute or conc. acid is used.
\[\ce{NH4OH + HCl->}\]
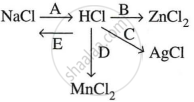
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.

Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.
Complete and balance the following reaction, state whether dilute or cone. acid is used.
\[\ce{NH4OH + HCl->}\]
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D and E.