Advertisements
Advertisements
Questions
State Faraday’s Laws of electrolysis.
State and explain Faraday’s laws of electrolysis.
What is Faraday’s first law of electrolysis?
Advertisements
Solution 1
Faraday’s laws of electrolysis:
- First law: The mass of the substance (M) liberated at an electrode during electrolysis is directly proportional to the quantity of charge (Q) passed through the cell, i.e., M ∝ Q.
- Second law: When the same quantity of charge is passed through the solutions of different electrolytes, the amount of substances liberated at the respective electrodes are directly proportional to their electrochemical equivalents, i.e., M ∝ Z.
Solution 2
Faraday’s first law of electrolysis: Faraday’s first law of electrolysis may be stated as follows.
The amount of substance liberated (or deposited) at a particular electrode during electrolysis is directly proportional to the quantity of electricity (total charge) passed in the solution.
W ∝ Q
Since Charge = Current × Time
we have Q = I × t
where I is the current (in amperes) passed in the solution for t seconds. Hence,
W ∝ I × t
or, W = Z × I × t ...(i)
In the above equation, Z is a constant known as the electrochemical equivalent of the substance liberated at the electrode.
If I = 1 ampere and t = 1 s, we have
W = Z
Hence, electrochemical equivalent may be defined as the amount of substance liberated by a current of one ampere passed for one second.
Faraday’s second law of electrolysis: Faraday’s second law of electrolysis can be stated as follows.
When the same quantity of electricity is passed through the solutions of different electrolytes connected in series, the masses of the substances liberated (or deposited) at the electrodes are directly proportional to their equivalent masses.
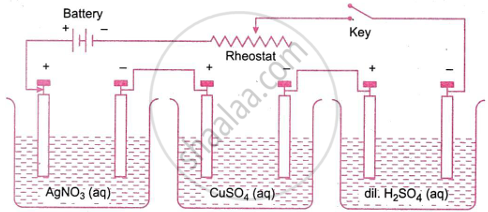
Suppose three voltameters connected in series contain aqueous solutions of silver nitrate, copper sulphate, and dilute sulphuric acid, respectively, as shown in Fig. On passing electric current, electrolysis takes place in all three cells, and the substances produced at the cathodes of these cells are Ag(s), Cu(s), and H2(g), respectively. If the same current is passed for the same time, it is found that
\[\frac{\text{Mass of silver deposited}}{\text{Mass of copper deposited}} = \frac{\text{Equivalent mass of silver}}{\text{Equivalent mass of copper}}\] and,
\[\frac{\text{Mass of hydrogen gas liberated}}{\text{Mass of silver deposited}} = \frac{\text{Equivalent mass of hydrogen}}{\text{Equivalent mass of silver}}\]
Notes
Students should refer to the answer according to their questions and preferred marks.
APPEARS IN
RELATED QUESTIONS
During electrolysis of molten sodium chloride, the time required to produce 0.1 mole of chlorine gas using a current of 3A is ___________.
While charging lead storage battery
Among the following cells
I) Leclanche cell
II) Nickel – Cadmium cell
III) Lead storage battery
IV) Mercury cell
Primary cells are:
In \[\ce{H2 - O2}\] fuel cell the reaction occurs at cathode is:
For the cell reaction
\[\ce{2Fe^{3+}_{( aq)} + 2l^-_{( aq)} -> 2Fe^{2+}_{( aq)} + l2_{( aq)}}\]
\[\ce{E^0_{cell}}\] = at 298 K. The standard Gibbs energy (∆G°) of the cell reactions is:
A current of 1.608A is passed through 250 mL of 0.5 M solution of copper sulphate for 50 minutes. Calculate the strength of Cu2+ after electrolysis assuming volume to be constant and the current efficiency is 100%.
Calculate the standard emf of the cell: \[\ce{Cd|Cd^{2+}||Cu^{2+}|Cu}\] and determine the cell reaction. The standard reduction potentials of Cu2+|Cu and Cd2+|Cd are 0.34 V and −0.40 volts respectively. Predict the feasibility of the cell reaction.
The same amount of electricity was passed through two separate electrolytic cells containing solutions of nickel nitrate and chromium nitrate respectively. If 2.935 g of Ni was deposited in the first cell. The amount of Cr deposited in the another cell?
Given: molar mass of Nickel and chromium are 58.74 and 52 gm−1 respectively.
8.2 × 1012 litres of water is available in a lake. A power reactor using the electrolysis of water in the lake produces electricity at the rate of 2 × 106 Cs−1 at an appropriate voltage. How many years would it like to completely electrolyse the water in the lake? Assume that there is no loss of water except due to electrolysis.
Explain the function of H2 – O2 fuel cell.
