Advertisements
Advertisements
Question
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
Advertisements
Solution 1
Acid dehydration of primary alcohols to ethers occurs by SN2 mechanism in which the nucleophilic attack of the alcohol molecule takes place on the protonated alcohol molecule.
\[\ce{CH3CH2CH2\overset{\bullet\bullet}{\underset{\bullet\bullet}{O}}H + CH3CH2CH2 - \overset{+}{\underset{\bullet\bullet}{O}}\overset{+}{H} ->[S_N2][-H+, -H2O] CH3CH2CH2 - O - CH2CH2CH3}\]
Under these conditions, secondary and tertiary alcohols give alkenes instead of ethers. There is no nucleophilic attack of the alcohol molecule on the protonated alcohol molecule. Instead, protonated secondary and tertiary alcohols lose a molecule of water to form stable 2° and 3° carbocations. These carbocations preferentially lose H+ to form alkenes.
\[\begin{array}{cc}
\ce{CH3}\phantom{...........................}\ce{CH3}\phantom{.....................}\ce{CH3}\phantom{}\\\
|\phantom{.................................}|\phantom{...........................}|\phantom{.....}\\
\ce{\underset{\underset{(2^\circ alcohol)}{Propan-2-ol}}{CH3 - CH - OH} ->[H^-] \underset{Protonated 2^\circ alcohol}{CH3 - CH - \overset{+}{O}H2} ->[][-H2O] \underset{2^\circ Carbocation}{CH3 - CH^+}}
\end{array}\]
\[\begin{array}{cc}
\ce{CH3}\phantom{...}\ce{CH3}\phantom{..............................}\ce{CH3}\phantom{...........}\\
|\phantom{.........}|\phantom{....................................}|\phantom{..............}\\
\ce{\underset{2-propoxy-2-propane}{CH3 - CH - O - CH - CH3} ->[CH3CHOCH3][-H^+] CH3 - CH^+ ->[][-H^+] \underset{Propene}{CH3 - CH = CH2}}
\end{array}\]
Similarly, 3° alcohols give alkenes instead of ethers.
\[\ce{\underset{\underset{(3^\circ alcohol)}{2-Methylpropan-2-ol}}{(CH3)3C - OH} ->[H+] \underset{\underset{(3^\circ alcohol)}{Protonated 2-methylpropan-2-ol}}{(CH3)3C - \overset{+}{O}H2} ->[][-H2O] \underset{3^\circ butyl carbocation}{(CH3)3C^+}}\]
\[\begin{array}{cc}
\phantom{.......}\ce{CH3}\phantom{.}\ce{CH3}\phantom{....................................}\ce{CH3}\phantom{..................}\ce{CH3}\\
\phantom{.....}|\phantom{......}|\phantom{..........................................}|\phantom{.........................}|\\
\ce{CH3 - C - O - C - CH3 ->[(CH3)3COH][-H^+] CH3 - C^+ ->[][-H^+] \underset{2-Methylprop-1-ene}{CH3 - CH = CH2}}\\
|\phantom{.......}|\phantom{..........................................}|\phantom{......................}\\
\ce{\underset{2-Methyl-2-(2-methyl-2-propoxy)propane}{\phantom{..}CH3\phantom{..}CH3}}\phantom{...............}\ce{\underset{3^\circ butyl carbocation}{CH3}}\phantom{............................}
\end{array}\]
Solution 2
Consider the reaction between propan-2-ol molecules in the presence of an acid.
\[\begin{array}{cc}
\phantom{..............}\ce{CH3}\phantom{...........................}\ce{CH3}\\
\phantom{..........}|\phantom{.................................}|\\
\ce{CH3 - CH - OH ->[H+] CH3 - \underset{+}{C}H}\\
\end{array}\]
If an ether is to be formed, another alcohol molecule must carry out a nucleophilic attack on the carbocation as:
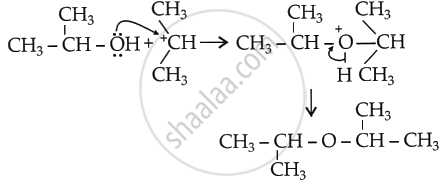
However, this does not happen because of:
- the steric hindrance around the carbocation, and
- the bulky size of the nucleophile would further cause crowding.
As a result, the carbocation prefers to lose a proton, forming an alkene.
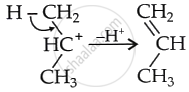
For the same reason, 3° alcohols in the presence of acid do not form ethers since 3° alcohols are even more sterically hindered than 2° alcohols.
APPEARS IN
RELATED QUESTIONS
Explain the mechanism of the following reaction:

How do you convert the following :
Phenol to anisole
Write the reactions of Williamson synthesis of 2-ethoxy-3-methylpentane starting from ethanol and 3-methylpentan-2-ol.
Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
Dehydration of alcohol to ethers is catalysed by:
Explain why low molecular mass alcohols are soluble in water.
The Williamson ether synthesis produces ethers by reacting an
Identify the following named reaction
\[\ce{C2H5Br ->[NaOCH3] C2H5OCH3}\]
Williamson's synthesis of ether is an example of ______.
Which of the following reactions are feasible?
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Write the names of reagents and equations for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Identify the product (s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Consider the following reaction:
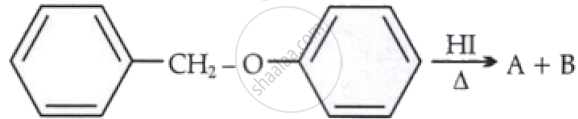
Identify products A and B.
