Advertisements
Advertisements
Question
How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of this reaction.
Advertisements
Solution
\[\ce{\underset{Propanol}{CH3CH2CH2OH} ->[?] \underset{Propoxypropane}{(CH3CH2CH2)2O}}\]
This reaction can be brought about as:
\[\ce{\underset{Propanol}{CH3CH2CH2OH} + SOCl2 -> CH3CH2CH2Cl}\]
\[\ce{2CH3CH2CH2CH2OH + \underset{(Metal)}{2Na} -> 2CH3CH2CH2CH2\overset{-}{O}N\overset{+}{a} + H2}\]
\[\ce{\underset{1-Chloropropane}{CH3CH2CH2Cl} + \underset{Sodium propoxide}{CH3CH2CH2\overset{-}{O}}N\overset{+}{a} -> \underset{Propoxypropane}{(CH3CH2CH2)2O}}\]
Mechanism: SN2 attack of propoxide on the halide.

APPEARS IN
RELATED QUESTIONS
Explain the mechanism of the following reaction:

Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
Ethoxybenzene
Write the name of the reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
1-Methoxyethane
Explain why low molecular mass alcohols are soluble in water.
The Williamson ether synthesis produces ethers by reacting an
HBr reacts with \[\ce{CH2 = CH - OCH3}\] under anhydrous conditions at room temperature to give ______.
Which of the following reactions are feasible?
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of the reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagents and equations for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Identify the product(s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Short Answer Question.
Identify the product (s) is / are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction
Write the names of reagents and equations for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Short Answer Question.
Identify the product (s) is/are formed when 1 – methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
Give the structure and IUPAC name of the metamers of 2-methoxy propane.
Consider the following reaction:
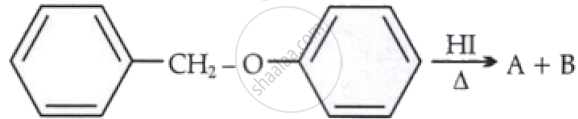
Identify products A and B.
