Advertisements
Advertisements
प्रश्न
How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of this reaction.
Advertisements
उत्तर
\[\ce{\underset{Propanol}{CH3CH2CH2OH} ->[?] \underset{Propoxypropane}{(CH3CH2CH2)2O}}\]
This reaction can be brought about as:
\[\ce{\underset{Propanol}{CH3CH2CH2OH} + SOCl2 -> CH3CH2CH2Cl}\]
\[\ce{2CH3CH2CH2CH2OH + \underset{(Metal)}{2Na} -> 2CH3CH2CH2CH2\overset{-}{O}N\overset{+}{a} + H2}\]
\[\ce{\underset{1-Chloropropane}{CH3CH2CH2Cl} + \underset{Sodium propoxide}{CH3CH2CH2\overset{-}{O}}N\overset{+}{a} -> \underset{Propoxypropane}{(CH3CH2CH2)2O}}\]
Mechanism: SN2 attack of propoxide on the halide.

APPEARS IN
संबंधित प्रश्न
Explain the mechanism of the following reaction:

How do you convert the following :
Phenol to anisole
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
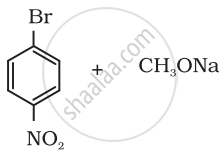 |
 |
| (i) | (ii) |
Write the name of the reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
1-Methoxyethane
Preparation of ethers by acid dehydration of secondary or tertiary alcohols is not a suitable method. Give reason.
Account for the following :
t-butyl chloride on heating with sodium methoxide gives 2-methylpropene instead of t-butyl methyl ether.
Williamson's synthesis of preparing dimethyl ether is a/an ____________.
Which of the following cannot be made by using Williamson Synthesis:
Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism.
Identify the following named reaction
\[\ce{C2H5Br ->[NaOCH3] C2H5OCH3}\]
Which of the following compounds gives a secondary alcohol upon reaction with methylmagnesium bromide?
Williamson's synthesis of ether is an example of ______.
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagents and equations for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of the reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride->[i)CH3MgBr][ii)H3O+]X ->[acidk2crp3]A}\]
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
