Advertisements
Advertisements
प्रश्न
Illustrate with examples the limitations of Williamson synthesis for the preparation of certain types of ethers.
Advertisements
उत्तर
The main limitation of Williamson's ether synthesis is that it cannot be used to prepare unsymmetrical ethers where the compound has tertiary or secondary alkyl groups. For example, a reaction between tert-butyl bromide and sodium methoxide yields an alkene.
\[\begin{array}{cc}
\ce{CH3}\phantom{..........................}\ce{CH2}\phantom{.........}\\
\phantom{.}|\phantom{..............................}||\phantom{............}\\
\ce{CH3 - C - Br + NaOCH3 -> CH3 - C + NaBr + CH3OH}\\
|\phantom{..............................}|\phantom{............}\\
\ce{\underset{tert-Butylbromide}{CH3}\phantom{..................}\ce{\underset{2-Methylpropene}{CH3}}}\phantom{..........}\
\end{array}\]
This is due to the formation of alkene and the dominance of the competing elimination reaction over SN2.
APPEARS IN
संबंधित प्रश्न
Write the equations involved in the following reactions : Williamson synthesis
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
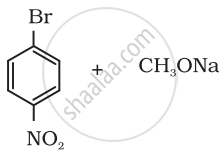 |
 |
| (i) | (ii) |
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of the reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
1-Methoxyethane
How is 1-propoxypropane synthesised from propan-1-ol? Write mechanism of this reaction.
Williamson's synthesis of preparing dimethyl ether is a/an ____________.
Explain why low molecular mass alcohols are soluble in water.
Name the suitable alcohol and reagent, from which 2-Chloro-2-methyl propane can be prepared.
Write the equations for the following reaction:
Tert butyl chloride is treated with sodium ethoxide.
Williamson's synthesis of ether is an example of ______.
Write the mechanism of the following reaction:
\[\ce{2CH3CH2OH ->[H^+][413 K] CH3-CH2-O-CH2-CH3 + H2O}\]
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Identify the product (s) is/are formed when 1-methoxy propane is heated with excess HI. Name the mechanism involved in the reaction.
What will be the product (X and A)for the following reaction
\[\ce{acetylchloride->[i)CH3MgBr][ii)H3O+]X ->[acidk2crp3]A}\]
Write the names of reagents and equations for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Give the structure and IUPAC name of metamers of 2-methoxy propane
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
