Advertisements
Advertisements
प्रश्न
Write the equations involved in the following reactions : Williamson synthesis
Advertisements
उत्तर १

उत्तर २
Williamson synthesis: It involves the treatment of an alkyl halide with a suitable sodium alkoxide. The sodium alkoxide needed for the purpose is prepared by the action of sodium on a suitable alcohol. The reaction invlolves the nucleophillic displacement (substitution) of the halide ion from the alkyl halide by the alkoxide ion by SN2 mechanism.
R-OH +2Na → 2R’-O-Na+ + H2
R’-O-Na+ + R-X → R’-O-R +Na+ X
Ether
The second reaction is the substitution nucleophillic bimolecular (SN2) reaction.
For example:

APPEARS IN
संबंधित प्रश्न
How do you convert the following :
Phenol to anisole
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
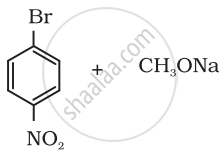 |
 |
| (i) | (ii) |
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
Ethoxybenzene
Explain the following with an example.
Williamson ether synthesis
Which of the following cannot be made by using Williamson Synthesis:
Explain why low molecular mass alcohols are soluble in water.
Williamson's synthesis of ether is an example of ______.
Which of the following reactions are feasible?
Write the name of the reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
