Advertisements
Advertisements
प्रश्न
Write the equations involved in the following reactions : Williamson synthesis
Advertisements
उत्तर १

उत्तर २
Williamson synthesis: It involves the treatment of an alkyl halide with a suitable sodium alkoxide. The sodium alkoxide needed for the purpose is prepared by the action of sodium on a suitable alcohol. The reaction invlolves the nucleophillic displacement (substitution) of the halide ion from the alkyl halide by the alkoxide ion by SN2 mechanism.
R-OH +2Na → 2R’-O-Na+ + H2
R’-O-Na+ + R-X → R’-O-R +Na+ X
Ether
The second reaction is the substitution nucleophillic bimolecular (SN2) reaction.
For example:

APPEARS IN
संबंधित प्रश्न
Preparation of alcohols from alkenes involves the electrophilic attack on alkene carbon atom. Explain its mechanism.
The Williamson ether synthesis produces ethers by reacting an
Identify the following named reaction
\[\ce{C2H5Br ->[NaOCH3] C2H5OCH3}\]
What is metamerism? Give the structure and IUPAC name of metamers of 2-methoxy propane.
Write the name of the reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Predict the major product, when 2-methyl but -2-ene is converted into an alcohol in the following method.
Acid catalysed hydration
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ether by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Give the structure and IUPAC name of the metamers of 2-methoxy propane.
Consider the following reaction:
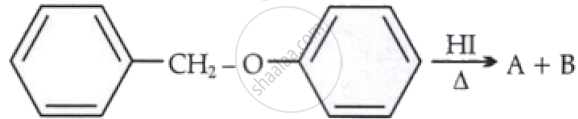
Identify products A and B.
