Advertisements
Advertisements
प्रश्न
Williamson's synthesis of preparing dimethyl ether is a/an ____________.
पर्याय
SN1 reactions
SN2 reaction
electrophilic addition
electrophilic substitution
Advertisements
उत्तर
Williamson's synthesis of preparing dimethyl ether is a/an SN2 reaction.
APPEARS IN
संबंधित प्रश्न
Which of the following is an appropriate set of reactants for the preparation of 1-methoxy-4-nitrobenzene and why?
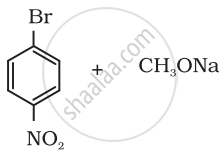 |
 |
| (i) | (ii) |
Write the name of the reagent and the equation for the preparation of the following ether by Williamson’s synthesis:
Ethoxybenzene
Explain why low molecular mass alcohols are soluble in water.
Williamson's synthesis of ether is an example of ______.
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagents and equations for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Write the name of reagent and equation for the preparation of the following ethers by Williamson’s synthesis:
2-Methoxy-2-methylpropane
Give the structure and IUPAC name of metamers of 2-methoxy propane
Consider the following reaction:
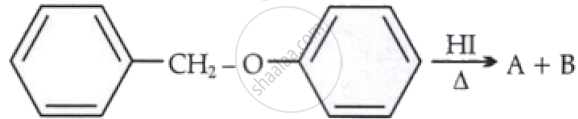
Identify products A and B.
