Advertisements
Advertisements
Question
Obtain an expression for de-Broglie wavelength of wave associated with material particles. The photoelectric work function for metal is 4.2 eV. Find the threshold wavelength.
Advertisements
Solution
De-Broglie equated the energy equation of Plank (wave nature) and Einstein (particle nature) such that,
E = hv (Plank energy relation)
E = mc2 (Einsteins mass-energy relation)
Where,
E = energy associated with the particle
h = planks constant
v = frequency associated with the particle
m = moss of the particle
c = speed of light
After equating equations (1) and (2) we get:
hv = mc2
`"h" "c"/lambda = "mc"^2 ...(because "v" = "c"/lambda)`
`lambda = "hc"/"mc"^2 = "h"/"mc"`
If the particle is moving with velocity 'v' then equation (3) becomes,
`lambda = "h"/"mv"`
The wavelength of the particle if energy is in electron volt is,
`lambda = (12,400 Å)/"E"`
Where E should be in eV.
After substituting the value of E i.e. 4.2 eV in equation (4) we get:
`lambda = (12,400 Å)/4.2`
= 2952.38 Å
Hence, the threshold wavelength of the particle is 2952.38 Å.
APPEARS IN
RELATED QUESTIONS
State the importance of Davisson and Germer experiment.
What is the speed of a proton having de Broglie wavelength of 0.08 Å?
The de Broglie wavelengths associated with an electron and a proton are the same. What will be the ratio of
- their momenta
- their kinetic energies?
Two particles have the same de Broglie wavelength and one is moving four times as fast as the other. If the slower particle is an α-particle, what are the possibilities for the other particle?
Find the ratio of the de Broglie wavelengths of an electron and a proton when both are moving with the (a) same speed, (b) the same kinetic energy, and (c) the same momentum. State which of the two will have a longer wavelength in each case.
According to De-Broglie, the waves are associated with ______
An electron is accelerated through a potential of 120 V. Find its de Broglie wavelength.
Calculate De Broglie's wavelength of the bullet moving with speed 90m/sec and having a mass of 5 gm.
The momentum of a photon of energy 1 MeV in kg m/s will be ______
The de Broglie wavelength associated with photon is, ____________.
If the radius of the innermost Bohr orbit is 0.53 Å, the radius of the 4th orbit is ______
What is the momentum of a photon having frequency 1.5 x 1013 Hz?
A particle of charge q, mass m and energy E has de-Broglie wavelength `lambda.` For a particle of charge 2q, mass 2m and energy 2E, the de-Broglie wavelength is ____________.
If '`lambda_1`' and '`lambda_2`' are de-Broglie wavelengths for electrons in first and second Bohr orbits in hydrogen atom, then the ratio '`lambda_2`' to '`lambda_1`' is (E1 = -13.6 eV) ____________.
If the kinetic energy of a particle is increased to 16 times its previous value, the percentage change in the de-Broglie wavelength of the particle is ____________.
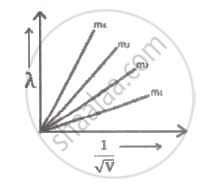
Graph shows the variation of de-Broglie wavelength `(lambda)` versus `1/sqrt"V"`, where 'V' is the accelerating potential for four particles carrying same charge but of masses m1 , m2, m3, m4. Which particle has a smaller mass?
A photon of wavelength 3315 Å falls on a photocathode and an electron of energy 3 x 10-19 J is ejected. The threshold wavelength of photon is [Planck's constant (h) = 6.63 x 10-34 J.s, velocity of light (c) = 3 x 108 m/s] ____________.
Explain de-Broglie wavelength.
A proton, a neutron, an electron and an α-particle have same energy. λp, λn, λe and λα are the de Broglie's wavelengths of proton, neutron, electron and α particle respectively, then choose the correct relation from the following :
The energy of an electron having de-Broglie wavelength `λ` is ______.
(h = Plank's constant, m = mass of electron)
An electron of mass m has de-Broglie wavelength λ when accelerated through potential difference V. When proton of mass M, is accelerated through potential difference 9V, the de-Broglie wavelength associated with it will be ______. (Assume that wavelength is determined at low voltage)
An electron is accelerated through a potential difference of 100 volts. Calculate de-Broglie wavelength in nm.
Calculate the de Broglie wavelength associated with an electron moving with a speed of `5 xx 10^6` m/s. `(m_e = 9.1 xx 10^(-31)kg)`
