Advertisements
Advertisements
प्रश्न
Obtain an expression for de-Broglie wavelength of wave associated with material particles. The photoelectric work function for metal is 4.2 eV. Find the threshold wavelength.
Advertisements
उत्तर
De-Broglie equated the energy equation of Plank (wave nature) and Einstein (particle nature) such that,
E = hv (Plank energy relation)
E = mc2 (Einsteins mass-energy relation)
Where,
E = energy associated with the particle
h = planks constant
v = frequency associated with the particle
m = moss of the particle
c = speed of light
After equating equations (1) and (2) we get:
hv = mc2
`"h" "c"/lambda = "mc"^2 ...(because "v" = "c"/lambda)`
`lambda = "hc"/"mc"^2 = "h"/"mc"`
If the particle is moving with velocity 'v' then equation (3) becomes,
`lambda = "h"/"mv"`
The wavelength of the particle if energy is in electron volt is,
`lambda = (12,400 Å)/"E"`
Where E should be in eV.
After substituting the value of E i.e. 4.2 eV in equation (4) we get:
`lambda = (12,400 Å)/4.2`
= 2952.38 Å
Hence, the threshold wavelength of the particle is 2952.38 Å.
APPEARS IN
संबंधित प्रश्न
What is the speed of a proton having de Broglie wavelength of 0.08 Å?
Explain what you understand by the de Broglie wavelength of an electron. Will an electron at rest have an associated de Broglie wavelength? Justify your answer.
The de Broglie wavelengths associated with an electron and a proton are the same. What will be the ratio of
- their momenta
- their kinetic energies?
Two particles have the same de Broglie wavelength and one is moving four times as fast as the other. If the slower particle is an α-particle, what are the possibilities for the other particle?
Find the ratio of the de Broglie wavelengths of an electron and a proton when both are moving with the (a) same speed, (b) the same kinetic energy, and (c) the same momentum. State which of the two will have a longer wavelength in each case.
An electron is accelerated through a potential of 120 V. Find its de Broglie wavelength.
Calculate De Broglie's wavelength of the bullet moving with speed 90m/sec and having a mass of 5 gm.
If the radius of the circular path and frequency of revolution of a particle of mass m are doubled, then the change in its kinetic energy will be (Ei and Ef are the initial and final kinetic energies of the particle respectively.)
A particle of charge q, mass m and energy E has de-Broglie wavelength `lambda.` For a particle of charge 2q, mass 2m and energy 2E, the de-Broglie wavelength is ____________.
If '`lambda_1`' and '`lambda_2`' are de-Broglie wavelengths for electrons in first and second Bohr orbits in hydrogen atom, then the ratio '`lambda_2`' to '`lambda_1`' is (E1 = -13.6 eV) ____________.
If the kinetic energy of a particle is increased to 16 times its previous value, the percentage change in the de-Broglie wavelength of the particle is ____________.
Graph shows the variation of de-Broglie wavelength `(lambda)` versus `1/sqrt"V"`, where 'V' is the accelerating potential for four particles carrying same charge but of masses m1 , m2, m3, m4. Which particle has a smaller mass?
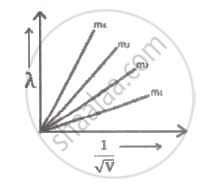
If the potential difference used to accelerate electrons is doubled, by what factor does the de-Broglie wavelength associated with the electrons change?
According to de-Broglie hypothesis, the ratio of wavelength of an electron and that of photon having same energy 'E' is (m = mass of electron, c = velocity of light) ____________.
A photon of wavelength 3315 Å falls on a photocathode and an electron of energy 3 x 10-19 J is ejected. The threshold wavelength of photon is [Planck's constant (h) = 6.63 x 10-34 J.s, velocity of light (c) = 3 x 108 m/s] ____________.
Explain de-Broglie wavelength.
The energy of an electron having de-Broglie wavelength `λ` is ______.
(h = Plank's constant, m = mass of electron)
An electron of mass m has de-Broglie wavelength λ when accelerated through potential difference V. When proton of mass M, is accelerated through potential difference 9V, the de-Broglie wavelength associated with it will be ______. (Assume that wavelength is determined at low voltage)
An electron is accelerated through a potential difference of 100 volts. Calculate de-Broglie wavelength in nm.
Calculate the de Broglie wavelength associated with an electron moving with a speed of `5 xx 10^6` m/s. `(m_e = 9.1 xx 10^(-31)kg)`
A photon and an electron have an equal energy ‘E’. The ratio of wavelength ‘λp’ of photon to that of electron ‘λe’ is proportional to ______.
