Advertisements
Advertisements
प्रश्न
The de Broglie wavelengths associated with an electron and a proton are the same. What will be the ratio of
- their momenta
- their kinetic energies?
The de-Broglie wavelengths associated with an electron and a proton are same. Calculate the ratio of their kinetic energies. (Given : mp = 1836 me)
The de-Broglie wavelengths associated with an electron and proton are same. Calculate the ratio of their momentum and kinetic energies.
Advertisements
उत्तर
Data: λ (electron) = λ (proton)
m (proton) = 1836 m (electron)
(i) λ = `h/p` As λ (electron) = λ (proton),
`(p("electron"))/(p("proton")) = 1`, where p denotes the magnitude of momentum.
(ii) Assuming v < < c,
KE = `1/2mv^2 = 1/2 (m^2 v^2)/m = p^2/(2m)`
`(KE("electron"))/(KE("proton")) = ((p_"electron")/(p_"proton"))^2 . (m_("proton"))/(m_("electron"))`
= 1 × 1836
= 1836 as p is the same for the electron and the proton.
APPEARS IN
संबंधित प्रश्न
An electron, a proton, an α-particle, and a hydrogen atom are moving with the same kinetic energy. The associated de Broglie wavelength will be longest for ______.
State the importance of Davisson and Germer experiment.
Two particles have the same de Broglie wavelength and one is moving four times as fast as the other. If the slower particle is an α-particle, what are the possibilities for the other particle?
Find the ratio of the de Broglie wavelengths of an electron and a proton when both are moving with the (a) same speed, (b) the same kinetic energy, and (c) the same momentum. State which of the two will have a longer wavelength in each case.
An electron is accelerated through a potential of 120 V. Find its de Broglie wavelength.
Calculate De Broglie's wavelength of the bullet moving with speed 90m/sec and having a mass of 5 gm.
Explain De Broglie’s Hypothesis.
An electron of mass m and a photon have same energy E. The ratio of de-Broglie wavelengths associated with them is ( c being velocity of light) ______.
What is the momentum of a photon having frequency 1.5 x 1013 Hz?
If the radius of the circular path and frequency of revolution of a particle of mass m are doubled, then the change in its kinetic energy will be (Ei and Ef are the initial and final kinetic energies of the particle respectively.)
A particle of charge q, mass m and energy E has de-Broglie wavelength `lambda.` For a particle of charge 2q, mass 2m and energy 2E, the de-Broglie wavelength is ____________.
How much energy is imparted to an electron so that its de-Broglie wavelength reduces from 10-10 m to 0.5 × 10-10 m? (E =energy of electron)
The wavelength '`lambda`' of a photon and de-Broglie wavelength of an electron have same value. The ratio of energy of a photon to kinetic energy of electron is (m = mass of electron, c = velocity of light, h = Planck's constant) ____________.
If '`lambda_1`' and '`lambda_2`' are de-Broglie wavelengths for electrons in first and second Bohr orbits in hydrogen atom, then the ratio '`lambda_2`' to '`lambda_1`' is (E1 = -13.6 eV) ____________.
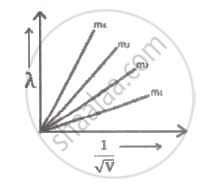
Graph shows the variation of de-Broglie wavelength `(lambda)` versus `1/sqrt"V"`, where 'V' is the accelerating potential for four particles carrying same charge but of masses m1 , m2, m3, m4. Which particle has a smaller mass?
Obtain an expression for de-Broglie wavelength of wave associated with material particles. The photoelectric work function for metal is 4.2 eV. Find the threshold wavelength.
The energy of an electron having de-Broglie wavelength `λ` is ______.
(h = Plank's constant, m = mass of electron)
An electron is accelerated through a potential difference of 100 volts. Calculate de-Broglie wavelength in nm.
Calculate the de Broglie wavelength associated with an electron moving with a speed of `5 xx 10^6` m/s. `(m_e = 9.1 xx 10^(-31)kg)`
A photon and an electron have an equal energy ‘E’. The ratio of wavelength ‘λp’ of photon to that of electron ‘λe’ is proportional to ______.
