Advertisements
Advertisements
Question
Name the following:
A metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Advertisements
Solution
K2Cr2O7 reacts with hydrochloric acid to give a coloured solution.
APPEARS IN
RELATED QUESTIONS
State your observations when Dilute Hydrochloric acid is added to Sodium thiosulphate.
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulfide, ethyne]
This gas has a characteristic rotten egg smell.
Give reason for the following:
An aqueous solution of chlorine in acidic in nature
Hint : Cl2 + H2O ⟶ HCl +HClO
Give three distinct test [apart from using an indicator] you would carry out with solution of HCl to illustrate the typical properties of an acid.
Convert two soluble metallic nitrates to insoluble metallic chlorides using dil. HCl.
What would happen if
Calcium oxide is used to dry hydrogen chloride.
Give a chemical test to distinguish between the following pair of compounds:
Sodium chloride solution and sodium nitrate solution.
Write observation:
Lead nitrate solution is mixed with dilute hydrochloric acid and heated.
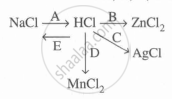
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
The ratio between the volumes occupied by 4.4 grams of carbon dioxide and 2 grams of hydrogen gas is ______.
