Advertisements
Advertisements
प्रश्न
Name the following:
A metallic oxide which reacts with hydrochloric acid to give a coloured solution.
Advertisements
उत्तर
K2Cr2O7 reacts with hydrochloric acid to give a coloured solution.
APPEARS IN
संबंधित प्रश्न
Give a chemical test to distinguish between the given pairs of chemicals:
Sodium chloride solution and Sodium nitrate solution
How will the action of dilute hydrochloric acid enable you to distinguish between sodium thiosulphate and sodium sulphite?
Convert two soluble metallic nitrates to insoluble metallic chlorides using dil. HCl.
State the composition of aqua regia. State which component is the oxidizing agent in aqua regia.
How will you distinguish between the two black samples, CuO and MnO2 with a chemical test?
Select from the list given (a to e) one substances in each case which matches the description given in parts (i) to (v). (Note : Each substance is used only one in the answer)
(a) Nitroso Iron (II) Sulphate
(b) Iron (III) chloride
(c) Chromium sulphate
(d) Lead (II) chloride
(e) Sodium chloride
(i) A compound which is deliquescent
(ii) A compound which is insoluble in cold water, but soluble in hot water
(iii) The compound responsible for the brown ring during the brown ring test of nitrate iron
(iv) A compound whose aqueous solution is neutral in nature
(v) The compound which is responsible for the green colouration when sulphur dioxide is passed through acidified potassium dichromate solution
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Write an equation for the reactions of hydrochloric acid on manganese (IV) oxide.
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.
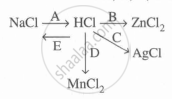
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
