Advertisements
Advertisements
Question
What would happen if
Calcium oxide is used to dry hydrogen chloride.
Advertisements
Solution
If calcium oxide is used to dry HCI, then it will react with HCI gas and result in the formation of calcium chloride and water.
CaO + 2HCl → CaCl2 + H2O
APPEARS IN
RELATED QUESTIONS
Potassium sulfite on reacting with hydrochloric acid releases _______ gas. (Cl2, SO2, H2S)
Write balanced chemical equations to show The behavior of H2SO4 as an acid when it reacts with Magnesium
State your observations when Dilute Hydrochloric acid is added to Sodium thiosulphate.
Select from the list the gas that matches the description given in case
[ammonia, ethane, hydrogen chloride, hydrogen sulfide, ethyne]
This gas has a characteristic rotten egg smell.
Name the gas that is produced in the given cases :
An action of dilute hydrochloride acid on sodium sulphide.
State which component is the oxidizing agent in aqua regia.
Write the balanced equation for the reaction of dilute hydrochloric acid with the following:
Iron
State which of the two - a solution of HCl in water or in toluene is an electrolyte. Explain.
Write an equation for the reaction of hydrochloric acid on manganese (IV) oxide.
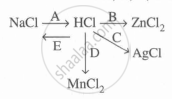
Study the flow chart and give balanced equations with conditions for the conversions A, B, C, D, and E.