Advertisements
Advertisements
Question
In what respects does a true solution differ from a colloidal solution ?
Advertisements
Solution
| Property | True Solution | Colloidal Solution |
| Size of the particles | < 1nm | 1 – 100 nm |
| Nature | Homogeneous | Heterogeneous |
| Tyndall effect | True Solution does not show Tyndall effect. | Colloidal solution shows Tyndall effect. |
| Separation | Cannot be done by filtration | Separation of colloidal solution cannot be done by filtration. However, it is possible by centrifugation process. |
APPEARS IN
RELATED QUESTIONS
Define the following term of Matter .
Name a non-metal which is a good conductor of electricity.
Name one element, one compound and one mixture.
Give reason why copper metal is used for making electric wires.
Name a metal which has a low melting point.
Classify the following into elements, compounds and mixtures :
arble, Air, Gold, Brass, Sand, Diamond, Graphite, Petroleum, Common salt, Sea-water, Chalk
Which of the following are compounds ?
- CO
- No
- NO
- Co
“Is malleable and ductile” best describes :
How much water should be added to 15 grams of salt to obtain 15 per cent salt solution ?
Fill in the following blank :
Milk is a .................... solution but vinegar is a .................... solution.
Classify the following into solutions, suspensions and colloids :
Soda-water, Milk, Brine, Blood, Ink, Smoke in air, Chalk water mixture, Milk of Magnesia, Shaving cream, Muddy river water.
State whether the following statement are true or false :
Alcohol can be separated from a mixture of alcohol and water by a separating funnel.
Which technique is used in a washing machine to squeeze out water from wet clothes while drying ?
The liquid air has three components X, Y and Z whose boiling points are : –186°C, –183°C and –196°C,
respectively. When liquid air is fed into a tall fractional distillation column from near its bottom and warmed
up slowly :
(a) Which component will be collected from near the bottom of the fraction distillation column ? Why ?
(b) Which component will be collected from the top part of the fractional distillation column ? Why ?
(c) Which component will be collected from the middle part of the fractional distillation column ? Why ?
(d) What could the component X, Y and Z be ?
There are three liquids A, B and C, all having different densities and different boiling points. Liquids A and C are organic in nature whereas liquid B is considered to be inorganic. When liquids A and B are put together in a container, they form a single layer. On the other hand, when, liquids B and C are mixed, they form two separate layers :
(a) Which process will you use to separate a mixture of A and B ?
(b) Which method will you use to separate a mixture of B and C ?
(c) Name the liquids which would behave like (i) A (ii) B and (iii) C.
Justify your answer.
| Figure 1 | Figure 2 | Figure 3 |
 |
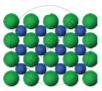 |
 |
The arrangement of particles in three different phases of matter is shown above.
- Which state is represented by Fig. 1?
- In which state will the inter-particle attraction be maximum?
- Which one of them cannot be contained in an open vessel?
- Which one can take the shape of its container?
Name the metals that are used in jewellery.
A teabag placed in a cup of hot water will diffuse into the water.
Write scientific reason.
Lemon sherbat has sweet, sour and salty taste and it can be poured in a glass.
