Advertisements
Advertisements
Question
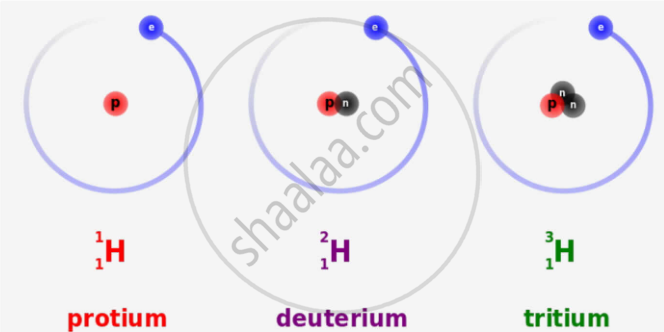
In what respects do the three isotopes of hydrogen differ? Give their structures.
One Line Answer
Advertisements
Solution
The three isotopes differ only due to their mass number which is respectively 1, 2 and 3 and named protium, deuterium and tritium.
shaalaa.com
Is there an error in this question or solution?
APPEARS IN
RELATED QUESTIONS
Write the names of `("NH"_4)_2"SO"_4` compounds:
Write the names of `"Ca"("NO"_3)_2` compounds :
Define: Chemical bond
An atom has 13 protons and 14 neutrons.
What is the atomic number of the atom ?
Fill in the blank
A charged particle is called an _____
Write the full form of IUPAC.
One nm is equal to :
Assertion: An atom is electrically neutral.
Reason: Atoms have equal number of protons and electrons.
The smallest unit of an element is ______.
What is the fundamental particle of an element?
