Advertisements
Advertisements
प्रश्न
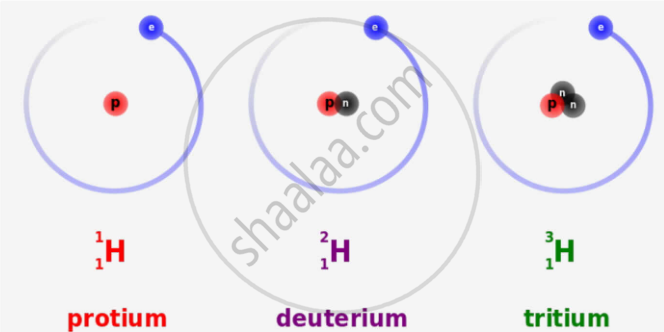
In what respects do the three isotopes of hydrogen differ? Give their structures.
Advertisements
उत्तर
The three isotopes differ only due to their mass number which is respectively 1, 2 and 3 and named protium, deuterium and tritium.
APPEARS IN
संबंधित प्रश्न
TRUE \ FALSE
Neils Bohr gave the theory to explain the arrangement of protons in an atom.
Define: Mass number
What is the maximum number of electrons present in:
1. first shell
2. second shell
3. third shell and
4. fourth shell
Write electronic configuration of oxygen, fluorine, sodium, silicon, argon.
A certain particle ‘X’ has 17 protons, 18 electrons and 20 neutrons.
Does the particle have an excess of electrons or a deficiency of electrons ?
Electron, proton, neutron are the types of ______ in an atom.
The smaller particles found in the atom is called ______.
Which of the following statements made by Dalton has not undergone any change?
In the nucleus of `""_20"Ca"^40`, there are ______
Atoms of one element can be transmuted into atoms of other elements by ______
