Advertisements
Advertisements
Question
Identify the part of the electromagnetic spectrum which is:
produced by bombarding a metal target by high speed electrons.
Advertisements
Solution
X-rays are produced by bombarding a metal target by high speed electrons.
APPEARS IN
RELATED QUESTIONS
Give a reason for the following:
Long-distance radio broadcasts use short-wave bands. Why?
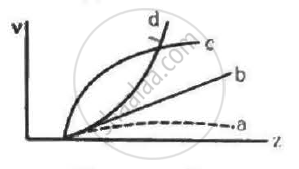
Frequencies of Kα X-rays of different materials are measured. Which one of the graphs in the figure may represent the relation between the frequency v and the atomic number Z ?
For harder X-rays,
(a) the wavelength is higher
(b) the intensity is higher
(c) the frequency is higher
(d) the photon energy is higher.
The Kα and Kβ X-rays of molybdenum have wavelengths 0.71 A and 0.63 A respectively. Find the wavelength of Lα X-ray of molybdenum.
The energy of a silver atom with a vacancy in K shell is 25.31 keV, in L shell is 3.56 keV and in M shell is 0.530 keV higher than the energy of the atom with no vacancy. Find the frequency of Kα, Kβ and Lα X-rays of silver.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
The distance between the cathode (filament) and the target in an X-ray tube is 1.5 m. If the cutoff wavelength is 30 pm, find the electric field between the cathode and the target.
(Use Planck constant h = 6.63 × 10-34 Js= 4.14 × 10-15 eVs, speed of light c = 3 × 108 m/s.)
Name the scientist who discovered Infra-red waves
Two waves A and B have wavelength 0.01 Å and 9000 Å respectively.
Name the two waves. compare the speeds of these waves when they travel in vacuum.
Identify the part of the electromagnetic spectrum which:
- produces the heating effect.
- is absorbed by the ozone layer in the atmosphere.
- is used for studying crystal structure.
Write any one method of the production of each of the above radiations.
